Abstract
The network of enzymes that contribute to the signal transduction of extracellular factors in pancreatic cancer is ever increasing. The classical Raf-MEK-ERK signaling cascade plays a crucial role in the regulation of apoptosis, proliferation, and metastasis of pancreatic cancer. Phosphatidylinositide-3-kinase also contributes to growth and prevents apoptosis in pancreatic cancer cells, acting in part via its downstream targets, PKB/AKT and the FRAP/p70s6k signaling complex. Recently, members of the PKC family of serine threonine kinases have emerged as novel modulators of transformation and cell cycle progression of pancreatic cancers. The novel PKD family of serine threonine kinases has just been detected in pancreatic cancer and awaits its functional characterization in these tumors.
Similar content being viewed by others
References
Yip-Schneider MT, Lin A, Marshall MS. Pancreatic tumor cells with mutant K-ras suppress ERK activity by MEK-dependent induction of MAP kinase phosphatase-2. Biochem Biophys Res Commun 2001;280:992–997.
Kornmann M, Arber N, Korc M. Inhibition of basal and mitogen-stimulated pancreatic cancer cell growth by cyclin D1 antisense is associated with loss of tumorigenicity and potentiation of cytotoxicity to cisplatinum. J Clin Invest 1998;101:344–352.
Wagner M, Lopez ME, Cahn M, Korc M. Suppression of fibroblast growth factor receptor signaling inhibits pancreatic cancer cell growth in vitro and in vivo. Gastroenterology 1998;114:798–807.
Seufferlein T, Van Lint J, Liptay S, Adler G, Schmid RM. Transforming growth factor alpha activates Ha-ras in human pancreatic cancer cells with Ki-ras mutations. Gastroenterology 1999;116:1441–1452.
Giehl K, Skripczynski B, Mansard A, Menke A, Gierschik P. Growth factor-dependent activation of the Ras-Raf-MEKMAPK pathway in the human pancreatic carcinoma cell line PANC-1 carrying activated K-ras: implications for cell proliferation and cell migration. Oncogene 2000;19:2930–2942.
Boucher MJ, Morisset J, Vachon PH, Reed JC, Laine J, Rivard N, MEK/ERK signalling pahway regulates the expression of Bcl-2, Bcl-X(L), and Mcl-1 and promotes survival of human pancreatic cancer cells. J Cell Biochem 2000;79:355–369.
Boucher MJ, Duchesne C, Laine J, Morisset J, Rivard N. cAMP protection of pancreatic cancer cells against apoptosis induced by ERK inhibition. Biochem Biophys Res Commun 2001;285:207–216.
Ellenrieder V, Hendler SF, Boeck W, et al. Transforming growth factor beta1 treatment leads to an epithelialmesenchymal transdifferentiation of pancreatic cancer cells requiring extracellular signal-regulated kinase 2 activation. Cancer Res 2001;61:4222–4228.
Su GH, Hilgers W, Shekher MC, et al. Alterations in pancreatic, biliary, and breast carcinomas support MKK4 as a genetically targeted tumor suppressor gene. Cancer Res 1998;58:2339–2342.
Ding XZ, Adrian TE. MEK/ERK-mediated proliferation is negatively regulated by p38 map kinase in the human pancreatic cancer cell line, Panc-1. Biochem Biophys Res Commun 2001;282:447–453.
Perugini RA, McDade TP, Vittimberga FJ Jr, Callery MP. Pancreatic cancer cell proliferation is phosphatidylinositol 3-kinase dependent. J Surg Res 2000;90:39–44.
Ng SSW, Tsa MS, Chow S, Hedley DW. Inhibition phosphatidylinositide 3-kinase enhances gemcitabine-induced apoptosis in human pancreatic cancer cells. Cancer Res 2000;60:5451–5455.
Ferrari S, Thomas G. S6 phosphorylation and the p70s6k/p85s6k. Crit Rev Bio Mol Biol 1994;29:385–413.
Price DJ, Grove JR, Calvo V, Avruch J, Bierer BE. Rapamycin-induced inhibition of the 70-kilodalton S6 protein kinase. Science 1992;257:973–977.
von Manteuffel SR, Gingras AC, Ming XF, Sonenberg N, Thomas G. 4E-BP1 phosphorylation is mediated by the FRAP-p70s6k pathway and is independent of mitogenactivated protein kinase. Proc Natl Acad Sci USA 1996;93:4076–4080.
Chou MM, Blenis J. The 70 kDa S6 kinase: regulation of a kinase with multiple roles in mitogenic signalling. Curr Opin Cell Biol 1995;7:806–814.
Rousseau D, Gingras AC, Pause A, Sonenberg N. The eIF4E-binding proteins 1 and 2 are negative regulators of cell growth. Oncogene 1996;13:2415–2420.
Grewe M, Gansauge F, Schmid RM, Adler G, Seufferlein T. Regulation of cell growth and cyclin D1 expression by the constitutively active FRAP-p70s6K pathway in human pancreatic cancer cells. Cancer Res 1999;59:3581–3587.
Shah SA, Potter MW, Ricciardi R, Perugini RA, Callery MP. FRAP-p70s6K signaling is required for pancreatic cancer cell proliferation. J Surg Res 2001;97:123–130.
Cheng JQ, Ruggeri B, Klein WM, et al. Amplification of AKT2 in human pancreatic cells and inhibition of AKT2 expression and tumorigenicity by antisense RNA. Proc Natl Acad Sci USA 1996;93:3636–3641.
Tanno S, Tanno S, Mitsuuchi Y, Altomare DA, Xiao GH, Testa JR. AKT activation up-regulates insulin-like growth factor I receptor expression and promotes invasiveness of human pancreatic cancer cells. Cancer Res 2001;61:589–593. (This paper shows that AKT regulates IGF-IR expression in human pancreatic cancer cells as a downstream target of src and suggests that active AKT promotes the invasiveness of pancreatic cancer cells through the up-regulation of IGF-IR expression.)
Hirata M, Tsuchida A, Iwao T, et al. Cholecystokinin regulates the invasiveness of human pancreatic cancer cell lines via protein kinase C pathway. Int J Oncol 1999;14:1129–1135.
Trauzold A, Werman H, Arlt A, et al. CD95 and TRAIL receptor-mediated activation of protein kinase C and NF-kappaB contributes to apoptosis resistance in ductal pancreatic adenocarcinoma cells. Oncogene 2001;20:4258–4269. (This manuscript demonstrates that protein kinase C protects pancreatic cancer cells from anti-CD95 and TRAIL-mediated apoptosis by inducing NFkappaB.)
Detjen KM, Brembeck FH, Welzel M, et al. Activation of protein kinase C alpha inhibits growth of pancreatic cancer cells via p21cip-mediated G(1) arrest. J Cell Sci 2000;113:3025–3035.
Denham DW, Fran MG, Denham W, et al. Directed antisense therapy confirms the role of protein kinase C-alpha in the tumorigenicity of pancreatic cancer. Surgery 1998;124:218–223.
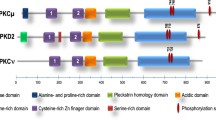
Van Lint J, Rykx A, Maeda Y, et al. Protein Kinase D: an intracellular traffic regulator on the move. Trends Cell Biol 2002;12:193–200. (This review provides a comprehensive overview on the current state of PKD-research.)
Guha S, Rey O, Rozengurt E. Neurotension Induces Protein Kinase C-dependent Protein Kinase D Activation and DNA Synthesis in Human Pancreatic Carcinoma Cell Line PANC-1. Cancer Res 2002;62:1632–1640.
Sturany S, Van Lint J, Mueller F, et al. Molecular cloning and characterization of the human protein kinase D2. J Biol Chem 2001;276:3310–3318.
Hayashi A, Seki N, Hattori A, Kozuma S, Saito T. PKCv, a new member of the protein kinase C family, composes a fourth subfamily with PKC. Biochim Biophys Acta 1999;1450:99–106.
Lutz MP, Esser IB, Flossmann-Kast BB, et al. Overexpression and activation of the tyrosine kinase src in human pancreatic carcinoma. Biochem Biophys Res Commun 1998;243:503–508.
Flossmann-Kast BB, Jehle PM, Hoeflich A, Adler G, Lutz MP. Src stimulates insulin-like growth factor I (IGF-I)-dependent cell proliferation by increasing IGF-I receptor number in human pancreatic carcinoma cells. Cancer Res 1998;58:3551–3554.
Sebolt-Leopold JS, Dudley DT, Herrera R, et al. Blockade of the MAP Kinase pathway suppresses growth of colon tumors in vivo. Nat Med 1999;5:810–816.
Alexandre J, Raymond E, Armand JP. Rapamycin and CCI-779. Bull Cancer 1999;86:808–811.
Guba M, von Breitenbuch P, Steinbauer M, et al. Rapamycin inhibits primary and metastatic tumor growth by antiangiogenesis: involvement of vascular endothelial growth factor. Nat Medicine 2002;8:128–135. (This paper describes the inhibition of metastatic tumor growth and angiogenesis in in vivo mouse models and the control of established tumors by the FRAP inhibitor, rapamycin. Rapmycin, which is also an immunosuppressant, may reduce the risk of recurrent or de novo cancer in transplant patients.)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seufferlein, T. Novel protein kinases in pancreatic cell growth and cancer. Int J Gastrointest Canc 31, 15–21 (2002). https://doi.org/10.1385/IJGC:31:1-3:15
Issue Date:
DOI: https://doi.org/10.1385/IJGC:31:1-3:15