Abstract
Background
The Controlling Nutritional Status (CONUT) score is an objective tool that is widely used to assess the nutritional status in patients, including those with cancer. The relationship between the CONUT score and prognosis in patients who have undergone hepatic resection has not been evaluated in a multi-institutional study.
Methods
Data were retrospectively collected for 2461 consecutive patients with hepatocellular carcinoma (HCC) who had undergone hepatic resection with curative intent at 13 institutions between January 2004 and December 2015. Patients were assigned to two groups: preoperative CONUT scores ≤ 3 (low CONUT score) and ≥ 4 (high CONUT score). Clinicopathological characteristics, surgical outcomes, and long-term survival were compared using propensity score matching analysis.
Results
Of the 2461 patients, 540 (21.9%) had high (≥ 4) and 1921 (78.1%) had low (≤ 3) preoperative CONUT scores. Overall, a high CONUT score was significantly associated with older age, female sex, low body mass index, low serum albumin, high serum total bilirubin, low lymphocyte count, low serum cholesterol, shorter prothrombin time, higher indocyanine green retention test at 15 min, Child–Pugh B (vs. A), liver cirrhosis, minor resection, shorter operation time, massive blood loss, blood transfusion, and postoperative complications. After propensity score matching, a higher CONUT score was significantly associated with poor overall survival (OS) and recurrence-free survival (RFS) using multivariate analysis.
Conclusions
This retrospective, multi-institutional analysis showed that, in patients who undergo curative hepatectomy for HCC, the preoperative CONUT score is predictive of worse OS and RFS, even after propensity score matching analysis.
Similar content being viewed by others
References
Llovcet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362:1907–17.
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557–76.
Alberino F, Gatta A, Amodio P et al. Nutrition and survival in patients with liver cirrhosis. Nutrition 2001;17:445–50.
Takenaka K, Kawahara N, Yamamoto K, et al. Results of 280 liver resections for hepatocellular carcinoma. Arch Surg. 1996;131:71–6.
Shimada M, Takenaka K, Gion T, et al. Prognosis of recurrent hepatocellular carcinoma: a 10-year surgical experience in Japan. Gastroenterology. 1996;111:720–6.
Shirabe K, Takeishi K, Taketomi A, Uchiyama H, Kayashima H, Maehara Y. Improvement of long-term outcomes in hepatitis C virus antibody-positive patients with hepatocellular carcinoma after hepatectomy in the modern era. World J Surg. 2011;35(5):1072–84.
Taketomi A, Kitagawa D, Itoh S, et al. Trends in morbidity and mortality after hepatic resection for hepatocellular carcinoma: an institute’s experience with 625 patients. J Am Coll Surg. 2007;204:580–7.
Yamashita Y, Taketomi A, Itoh S, et al. Longterm favorable results of limited hepatic resections for patients with hepatocellular carcinoma: 20 years of experience. J Am Coll Surg. 2007;205:19–26.
Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Br J Cancer. 2012;106(8):1439–45.
Chan AW, Chan SL, Wong GL, Wong VW, Chong CC, Lai PB, et al. Prognostic Nutritional Index (PNI) predicts tumor recurrence of very early/early stage hepatocellular carcinoma after surgical resection. Ann Surg Oncol. 2015;22(13):4138–48
de Ulibarri Perez JI, Gonzalez-Madrono A, de Villar NG, Gonzalez P, Gonzalez B, Mancha A, et al. CONUT: A tool for Controlling Nutritional Status. First validation in a hospital population. Nutr Hosp. 2005;20:38–45.
Harimoto N, Yoshizumi T, Sakata K, Nagatsu A, Motomura T, Itoh S, et al. Prognostic Significance of Preoperative Controlling Nutritional Status (CONUT) score in patients undergoing hepatic resection for hepatocellular carcinoma. World J Surg. 2017;41(11):2805–12.
Takagi K, Yagi T, Umeda Y, Shinoura S, Yoshida R, Nobuoka D, et al. Preoperative Controlling Nutritional Status (CONUT) score for assessment of prognosis following hepatectomy for hepatocellular carcinoma. World J Surg. 2017;41(9):2353–60.
Tokunaga R, Sakamoto Y, Nakagawa S, Ohuchi M, Izumi D, Kosumi K, et al. CONUT: a novel independent predictive score for colorectal cancer patients undergoing potentially curative resection. Int J Colorectal Dis. 2017;32(1):99–106.
Shoji F, Haratake N, Akamine T, Takamori S, Katsura M, Takada K, Toyokawa G, et al. The preoperative controlling nutritional status score predicts survival after curative surgery in patients with pathological stage i non-small cell lung cancer. Anticancer Res. 2017;37(2):741–7.
Kuroda D, Sawayama H, Kurashige J, Iwatsuki M, Eto T, Tokunaga R, et al. Controlling Nutritional Status (CONUT) score is a prognostic marker for gastric cancer patients after curative resection. Gastric Cancer. 2018;21(2):204–12.
Toyokawa G, Kozuma Y, Matsubara T, Haratake N, Takamori S, Akamine T, et al. Prognostic impact of controlling nutritional status score in resected lung squamous cell carcinoma. J Thorac Dis. 2017;9(9):2942–51.
Ishihara H, Kondo T, Yoshida K, Omae K, Takagi T, Iizuka J, et al. Preoperative controlling nutritional status (CONUT) score as a novel predictive biomarker of survival in patients with localized urothelial carcinoma of the upper urinary tract treated with radical nephroureterectomy. Urol Oncol. 2017;35(9):539.e9-539.
Miyata T, Yamashita YI, Higashi T, Taki K, Izumi D, Kosumi K, et al. The Prognostic Impact of Controlling Nutritional Status (CONUT) in intrahepatic cholangiocarcinoma following curative hepatectomy: a retrospective single institution study. World J Surg. 2018;42(4):1085–91.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Rahbari NN, Koch M, Mehabi A, et al. Portal triad clamping versus vascular exclusion for vascular control during hepatic resection: a systematic review and meta-analysis. J Gastrointest Surg. 2009;13:558–68.
Liver Cancer Study Group of Japan. General rules for the clinical and pathological study of primary liver cancer. Second English edition. Tokyo: Kanehara & Co.; 2003. pp. 34–35.
Abe T, Tashiro H, Kobayashi T, Hattori M, Kuroda S, Ohdan H. Glasgow Prognostic Score and prognosis after hepatectomy for hepatocellular carcinoma. World J Surg. 2017;41(7):1860–70.
Yoon YI, Kim KH, Kang SH, Kim WJ, Shin MH, Lee SK, et al. Pure laparoscopic versus open right hepatectomy for hepatocellular carcinoma in patients with cirrhosis: a propensity score matched analysis. Ann Surg. 2017;265(5):856–63.
de Ulibarri Perez JI, Femandez G, Rodriguez Salvanes F, Diaz Lopez AM. Nutritional screening control of clinical undernutrition with analytical parameters. Nutr Hosp. 2014; 29: 797–811.
Cengiz O, Kocer B, Sürmeli S, Santicky MJ, Soran A. Are pretreatment serum albumin and cholesterol levels prognostic tools in patients with colorectal carcinoma? Med Sci Monit. 2006;12(6):CR240-7.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860–7.
Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature 2008;454:436–44.
Yao W, He JC, Yang Y, Wang JM, Qian YW, Yang T, et al. The prognostic value of tumor-infiltrating lymphocytes in hepatocellular carcinoma: a systematic review and meta-analysis. Sci Rep. 2017;7(1):7525.
Harimoto N, Shirabe K, Yamashita YI et al. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg. 2013;100(11):1523–30.
Harimoto N, Yoshizumi T, Shimokawa M, Sakata K, Kimura K, Itoh S, et al. Sarcopenia is a poor prognostic factor following hepatic resection in patients aged 70 years and older with hepatocellular carcinoma. Hepatol Res. 2016;46(12):1247–55.
Acknowledgment
The authors thank Jodi Smith, Ph.D., from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Norifumi Harimoto, Tomoharu Yoshizumi, Shoichi Inokuchi, Shinji Itoh, Eisuke Adachi, Yasuharu Ikeda, Hideaki Uchiyama, Tohru Utsunomiya, Kiyoshi Kajiyama, Koichi Kimura, Fumiaki Kishihara, Keishi Sugimachi, Eiji Tsujita, Mizuki Ninomiya, Kengo Fukuzawa, Takashi Maeda, Ken Shirabe, and Yoshihiko Maehara have no conflicts of interest or financial support to declare.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Electronic Supplementary Fig.
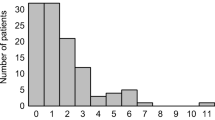
1 Receiver operating characteristics (ROC) curves of the preoperative CONUT score were analyzed, with (a) overall survival (OS), (b) cancer-specific survival (CSS), and (c) recurrence-free survival (RFS) predicted by comparing the areas under the curve (AUCs). Electronic Supplementary Fig. 2 Histograms of the area of CONUT scores for all patients. The histograms of all populations were normally distributed. Electronic Supplementary Fig. 3 Kaplan–Meier analysis of (a) overall survival and (b) recurrence-free survival curves in patients with CONUT scores ≥ 4 (thick black lines) and ≤ 3 (thin black lines). Both overall and recurrence-free survival were significantly poorer in patients with CONUT score ≥ 4. Electronic Supplementary Fig. 4 Kaplan–Meier analysis of (a) overall survival and (b) recurrence-free survival curves in patients with CONUT scores ≥ 4 (thick black lines) and ≤ 3 (thin black lines). Both overall and recurrence-free survival were significantly poorer in patients with CONUT score ≥ 4. Electronic Supplementary Fig. 5 Kaplan–Meier analysis of (a) overall survival and (b) recurrence-free survival curves in patients with CONUT scores ≥ 4 (thick black lines) and ≤ 3 (thin black lines) in the low-risk subgroups, meaning thepatients with no liver cirrhosis, a single tumor, and tumor size less than 5 cm. Both overall and recurrence-free survival were significantly poorer in patients with CONUT score ≥ 4. (PPTX 146 kb)
Rights and permissions
About this article
Cite this article
Harimoto, N., Yoshizumi, T., Inokuchi, S. et al. Prognostic Significance of Preoperative Controlling Nutritional Status (CONUT) Score in Patients Undergoing Hepatic Resection for Hepatocellular Carcinoma: A Multi-institutional Study. Ann Surg Oncol 25, 3316–3323 (2018). https://doi.org/10.1245/s10434-018-6672-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6672-6