Abstract
Background
We postulated that the worse prognosis of melanoma with advancing age reflected more aggressive tumor biology and that in younger patients the prognosis would be more favorable.
Materials and Methods
The expanded AJCC melanoma staging database contained 11,088 patients with complete data for analysis, including mitotic rate.
Results
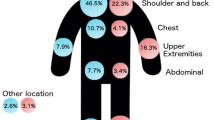
With increasing age by decade, primary melanomas were thicker, exhibited higher mitotic rates, and were more likely to be ulcerated. In a multivariate analysis of patients with localized melanoma, thickness and ulceration were highly significant predictors of outcome at all decades of life (except for patients younger than 20 years). Mitotic rate was significantly predictive in all age groups except patients <20 and >80 years. For patients with stage III melanoma, there were four independent variables associated with patient survival: number of nodal metastases, patient age, ulceration, and mitotic rate. Patients younger than 20 years of age had primary tumors with slightly more aggressive features, a higher incidence of sentinel lymph node metastasis, but, paradoxically, more favorable survival than all other age groups. In contrast, patients >70 years old had primary melanomas with the most aggressive prognostic features, were more likely to be head and neck primaries, and were associated with a higher mortality rate than the other age groups. Surprisingly, however, these patients had a lower rate of sentinel lymph node metastasis per T stage. Among patients between the two age extremes, clinicopathologic features and survival tended to be more homogeneous.
Conclusions
Melanomas in patients at the extremes of age have a distinct natural history.



Similar content being viewed by others
References
Balch CM, Gershenwald JE, Soong SJ, Thompson JF, Atkins MB, Byrd DR, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199–206.
Balch CM, Gershenwald JE, Soong SJ, Thompson JF, Ding S, Byrd DR, et al. Multivariate analysis of prognostic factors among 2,313 patients with stage III melanoma: comparison of nodal micrometastases versus macrometastases. J Clin Oncol. 2010;28:2452–9.
Balch CM, Soong SJ, Gershenwald JE, Thompson JF, Reintgen DS, Cascinelli N, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2001;19:3622–34.
Balch CM, Soong SJ, Milton GW, Shaw HM, McGovern VJ, Murad TM, et al. A comparison of prognostic factors and surgical results in 1,786 patients with localized (stage I) melanoma treated in Alabama, USA, and New South Wales, Australia. Ann Surg. 1982;196:677–84.
Balch CM, Soong SJ, Murad TM, Ingalls AL, Maddox WA. A multifactorial analysis of melanoma. II. Prognostic factors in patients with stage I (localized) melanoma. Surgery. 1979;86:343–51.
Balch CM, Soong SJ, Murad TM, Ingalls AL, Maddox WA. A multifactorial analysis of melanoma: III. Prognostic factors in melanoma patients with lymph node metastases (stage II). Ann Surg. 1981;193:377–88.
Ferrone CR, Panageas KS, Busam K, Brady MS, Coit DG. Multivariate prognostic model for patients with thick cutaneous melanoma: importance of sentinel lymph node status. Ann Surg Oncol. 2002;9:637–45.
Kretschmer L, Starz H, Thoms KM, Satzger I, Völker B, Jung K, et al. Age as a key factor influencing metastasizing patterns and disease-specific survival after sentinel lymph node biopsy for cutaneous melanoma. Int J Cancer. 2011;129:1435–42.
Lasithiotakis K, Leiter U, Meier F, Eigentler T, Metzler G, Moehrle M, et al. Age and gender are significant independent predictors of survival in primary cutaneous melanoma. Cancer. 2008;112:1795–804.
Macdonald JB, Dueck AC, Gray RJ, Wasif N, Swanson DL, Sekulic A, et al. Malignant melanoma in the elderly: different regional disease and poorer prognosis. J Cancer. 2011;2:538–43.
Thompson JF, Soong SJ, Balch CM, Gershenwald JE, Ding S, Coit DG, et al. Prognostic significance of mitotic rate in localized primary cutaneous melanoma: an analysis of patients in the multi-institutional American Joint Committee on Cancer melanoma staging database. J Clin Oncol. 2011;29:2199–205.
Wiener M, Acland KM, Shaw HM, Soong SJ, Lin HY, Chen DT, et al. Sentinel node positive melanoma patients: prediction and prognostic significance of nonsentinel node metastases and development of a survival tree model. Ann Surg Oncol. 2010;17:1995–2005.
Balch CM, Murad TM, Soong SJ, Ingalls AL, Halpern NB, Maddox WA. A multifactorial analysis of melanoma: prognostic histopathological features comparing Clark’s and Breslow’s staging methods. Ann Surg. 1978;188:732–42.
Caraco C, Marone U, Botti G, Celentano E, Lastoria S, Mozzillo N. Age as predictor in patients with cutaneous melanoma submitted to sentinel lymph node biopsy. Eur J Surg Oncol. 2006;32:970–3.
Egger ME, Callender GG, McMasters KM, Ross MI, Martin RC 2nd, Edwards MJ, et al. Diversity of stage III melanoma in the era of sentinel lymph node biopsy. Ann Surg Oncol. 2013;20:956–63.
Balch CM, Gershenwald JE, Atkins MB, et al. Melanoma of the skin. In Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti III A, eds. AJCC Cancer Staging Manual. 7th ed. New York: Springer; 2009:325–44.
Cox DR. Regression models and life-tables. J R Stat Soc Series B Stat Methodol. 1972;34:187–220.
Jemal A, Saraiya M, Patel P, Cherala SS, Barnholtz-Sloan J, Kim J, et al. Recent trends in cutaneous melanoma incidence and death rates in the United States, 1992–2006. J Am Acad Dermatol. 2011;65:S17–25
Lange JR, Palis BE, Chang DC, Soong SJ, Balch CM. Melanoma in children and teenagers: an analysis of patients from the National Cancer Data Base. J Clin Oncol. 2007;25:1363–8.
Strouse JJ, Fears TR, Tucker MA, Wayne AS. Pediatric melanoma: risk factor and survival analysis of the surveillance, epidemiology and end results database. J Clin Oncol. 2005;23:4735–41.
Testori A, Soteldo J, Sances D, Mazzarol G, Trifirò G, Zonta M, et al. Cutaneous melanoma in the elderly. Melanoma Res. 2009;19:125–34.
Tsai S, Balch C, Lange J. Epidemiology and treatment of melanoma in elderly patients. Nat Rev Clin Oncol. 2010;7:148–52.
Simard EP, Ward EM, Siegel R, Jemal A. Cancers with increasing incidence trends in the United States: 1999 through 2008. CA Cancer J Clin. 2012. doi:10.3322/caac.20141
Geller AC, Miller DR, Annas GD, Demierre MF, Gilchrest BA, Koh HK. Melanoma incidence and mortality among US whites, 1969–1999. JAMA. 2002;288:1719–20.
Seidler AM, Pennie ML, Veledar E, Culler SD, Chen SC. Economic burden of melanoma in the elderly population: population-based analysis of the surveillance, epidemiology, and end results (seer)-medicare data. Arch Dermatol. 2010;146:249–56.
Chao C, Martin RC, 2nd, Ross MI, Reintgen DS, Edwards MJ, Noyes RD, et al. Correlation between prognostic factors and increasing age in melanoma. Ann Surg Oncol. 2004;11:259–64.
Pollack LA, Li J, Berkowitz Z, Weir HK, Wu XC, Ajani UA, et al. Melanoma survival in the United States, 1992 to 2005. J Am Acad Dermatol. 2011;65:S78–86.
Chagpar RB, Ross MI, Reintgen DS, Edwards MJ, Scoggins CR, Martin RC 2nd, et al. Factors associated with improved survival among young adult melanoma patients despite a greater incidence of sentinel lymph node metastasis. J Surg Res. 2007;143:164–8.
Sassen S, Shaw HM, Colman MH, Scolyer RA, Thompson JF. The complex relationships between sentinel node positivity, patient age, and primary tumor desmoplasia: analysis of 2,303 melanoma patients treated at a single center. Ann Surg Oncol. 2008;15:630–7.
Moore-Olufemi S, Herzog C, Warneke C, Gershenwald JE, Mansfield P, Ross M, et al. Outcomes in pediatric melanoma comparing prepubertal to adolescent pediatric patients. Ann Surg. 2011;253:1211–5.
Berk DR, LaBuz E, Dadras SS, Johnson DL, Swetter SM. Melanoma and melanocytic tumors of uncertain malignant potential in children, adolescents and young adults—the Stanford experience 1995–2008. Pediatr Dermatol. 2010;27:244–54.
Niakosari F, Kahn HJ, McCready D, Ghazarian D, Rotstein LE, Marks A, et al. Lymphatic invasion identified by monoclonal antibody D2-40, younger age, and ulceration: predictors of sentinel lymph node involvement in primary cutaneous melanoma. Arch Dermatol. 2008;144:462–7.
Paradela S, Fonseca E, Pita-Fernandez S, Kantrow SM, Diwan AH, Herzog C, et al. Prognostic factors for melanoma in children and adolescents: a clinicopathologic, single-center study of 137 patients. Cancer. 2010;116:4334–44.
Howman-Giles R, Shaw HM, Scolyer RA, Murali R, Wilmott J, McCarthy SW, et al. Sentinel lymph node biopsy in pediatric and adolescent cutaneous melanoma patients. Ann Surg Oncol. 2010;17:138–43.
Livestro DP, Kaine EM, Michaelson JS, Mihm MC, Haluska FG, Muzikansky A, et al. Melanoma in the young: Differences and similarities with adult melanoma—a case-matched controlled analysis. Cancer. 2007;110:614–24.
Busam KJ, Murali R, Pulitzer M, McCarthy SW, Thompson JF, Shaw HM, et al. Atypical spitzoid melanocytic tumors with positive sentinel lymph nodes in children and teenagers, and comparison with histologically unambiguous and lethal melanomas. Am J Surg Pathol. 2009;33:1386–95.
Meyers MO, Yeh JJ, Deal AM, Byerly FL, Woosley JT, Frank J, et al. Age and Breslow depth are associated with a positive sentinel lymph node in patients with cutaneous melanocytic tumors of uncertain malignant potential. J Am Coll Surg. 2010;211:744–8.
Daniotti M, Ferrari A, Frigerio S, Casieri P, Miselli F, Zucca E, et al. Cutaneous melanoma in childhood and adolescence shows frequent loss of INK4A and gain of KIT. J Invest Dermatol. 2009;129:1759–68.
Lewis KG. Trends in pediatric melanoma mortality in the United States, 1968 through 2004. Dermatol Surg. 2008;34:152–9.
Soong SJ, Ding S, Coit D, Balch CM, Gershenwald JE, Thompson JF, et al. Predicting survival outcome of localized melanoma: an electronic prediction tool based on the AJCC Melanoma Database. Ann Surg Oncol. 2010;17:2006–14.
Long GV, Menzies AM, Nagrial AM, Haydu LE, Hamilton AL, Mann GJ, et al. Prognostic and clinicopathologic associations of oncogenic BRAF in metastatic melanoma. J Clin Oncol. 2011;29:1239–46.
Acknowledgment
The study of the AJCC/UICC Melanoma Staging Committee was supported by a grant from the American Joint Committee on Cancer and by grants from the National Cancer Institute (P30 CA13148 at the University of Alabama at Birmingham and P50 CA93459 SPORE grant in melanoma at The University of Texas M.D. Anderson Cancer Center in Houston, TX). Three meetings held by the Committee were partially supported by an unrestricted educational grant from Schering-Plough (Kenilworth, NJ). The authors thank Ms. Gwen Berry at the John Wayne Cancer Institute for her editorial assistance.
Disclosure
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Seng-jaw Soong—deceased.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Balch, C.M., Soong, Sj., Gershenwald, J.E. et al. Age as a Prognostic Factor in Patients with Localized Melanoma and Regional Metastases. Ann Surg Oncol 20, 3961–3968 (2013). https://doi.org/10.1245/s10434-013-3100-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3100-9