Abstract
Background
The Tensin4 gene is involved in various biological events by mediating signal transduction. This study was designed to clarify its clinical significance in gastric cancer.
Methods
A total of 114 gastric cancer patients were enrolled in this study, and we obtained paired samples from tumor tissue and matched normal mucosa. Total RNA was extracted, and TNS4 mRNA expression was quantified using real-time reverse-transcription polymerase chain reaction. TNS4 protein expression was investigated by immunohistochemistry. Additionally, the relationship between TNS4 expression and clinicopathologic factors was statistically analyzed.
Results
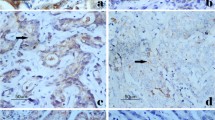
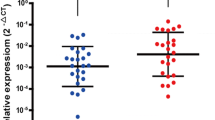
TNS4 mRNA expression was significantly higher in tumor tissue than in normal mucosa (p < 0.0001). Additional immunohistochemical analysis demonstrated that TNS4 protein was predominantly expressed in gastric cancer. Tumors with higher TNS4 mRNA expression showed histologically poorer grade (p < 0.02), deeper invasion into the serosa (p < 0.01), more positive lymph node metastasis (p < 0.02) or peritoneal dissemination (p < 0.05). Patients with high TNS4 mRNA expression tumor were more likely to have cancer-related death (p < 0.05). A Kaplan–Meier curve also demonstrated that patients with high TNS4 expression tumor had significantly poorer prognosis (p < 0.01) than those with low TNS4 expression tumor. In particular, the 2-year overall survival rates were 48.2% and 83.1%, respectively, a very significant difference. Multivariate analysis revealed that high TNS4 mRNA expression was an independent prognostic predictor (relative risk 1.41, 95% confidence interval 1.03–1.96, p = 0.03).
Conclusion
TNS4 was frequently overexpressed in gastric cancer, and tumors with high TNS4 mRNA expression showed biologically aggressive behavior. High TNS4 mRNA expression may be a novel prognostic predictor for those patients.




Similar content being viewed by others
References
Alberts SR, Cervantes A, van de Velde CJ. Gastric cancer: epidemiology, pathology and treatment. Ann Oncol 2003; 14:31–6
Roder DM. The epidemiology of gastric cancer. Gastric Cancer 2002; 5:5–11
Jemal A, Tiwari RC, Murray T, et al. Cancer Statistics, 2004. CA Cancer J Clin 2004; 54:8–29
Agochiya M, Brunton VG, Owens DW, et al. Increased dosage and amplification of the focal adhesion kinase gene in human cancer cells. Oncogene 1999; 18:5646–53
Chen H, Ishii A, Wong WK, et al. Molecular characterization of human tensin. Biochem J 2000; 351:403–11
Zhou MM, Ravichandran KS, Olejniczak EF, et al. Structure and ligand recognition of the phosphotyrosine binding domain of Shc. Nature 1995; 378:584–92
Davis S, Lu ML, Lo SH, et al. Presence of an SH2 domain in the actin-binding protein tensin. Science 1991; 252:712–5
Lo SH, Weisberg E, Chen LB. Tensin: A potential link between the cytoskeleton and signal transduction. Bioessays 1994; 16:817–23
Tice DA, Biscardi JS, Nickles AL, et al. Mechanism of biological synergy between cellular Src and epidermal growth factor receptor. Proc Natl Acad Sci USA 1999; 96:1415–20
Lo SH, An Q, Bao S, et al. Molecular cloning of chick cardiac muscle tensin. Full-length cDNA sequence, expression and characterization. J Biol Chem 1994; 269:22310–9
Kook S, Kim DH, Shim SR, et al. Caspase-dependent cleavage of tensin induces disruption of actin cytoskeleton during apoptosis. Biochem Biophys Res Commun 2003; 303:37–45
Cui Y, Liao Y, Lo SH. Epidermal growth factor modurates Tyrosine phosphorylation of a novel tensin family member, tensin3. Mol Cancer Res 2004; 2:225–32
Bockholt SM, Burridge K. Cell spreading on extracellular matrix proteins induces tyrosine phosphorylation of tensin. J Biol Chem 1993; 268:14565–7
Auger KR, Songyang Z, Lo SH, et al. Platelet-derived growth factor-induced formation of tensin and phosphoinositide 3-kinase complexes. J Biol Chem 1996; 271:23452–7
Lo S, Lo S., Huey Lo SH. Cleavage of cten by caspase-3 during apoptosis. Oncogene 2005; 24:4311–4
Sasaki H, Moriyama S, Mizuno K, et al. Cten mRNA expression was correlated with tumor progression in lung cancers. Lung Cancer 2003; 40:151–5
Sasaki H, Yukiue H, Kobayashi Y, et al. Cten mRNA expression is correlated with tumor progression in thymoma. Tumor Biol 2003; 24:271–4
Lo SH, Lo TB. A COOH-terminal Tensin-like protein with prostate restricted expression, is down-regulated in prostate cancer. Cancer Res 2002; 62:4217–21
Liao YC, Si L, deVere White RW, et al. The phosphotyrosine-independent interaction of DLC-1 and the SH2 domain of cten regulates focal adhesion localization and growth suppression activity of DLC-1. J Cell Biol 2007; 176:43–9
Mimori K, Mori M, Shiraishi T, et al. Clinical significance of tissue inhibitor of metalloproteinase expression in gastric carcinoma. Br J Cancer 1997; 76:531–6
Japanese Gastric Cancer Association: Japanese Classification of Gastric Cancer, 2nd English Edition. Gastric Cancer 1998; 1:10–24
Jass JR, Sobin LH, Watanabe H. The World Health Organization’s histologic classification of gastrointestinal tumors. A commentary on the second edition. Cancer 1990; 66:2162–7
Ogawa K, Utsunomiya T, Mimori K, et al. Clinical significance human kallikrein gene 6 messenger RNA expression in colorectal cancer. Clin Cancer Res 2005; 11:2889–93
Ohmachi T, Tanaka F, Mimori K, et al. Clinical significance of TROP2 expression in colorectal cancer. Clin Cancer Res 2006; 12:3057–63
Sasaki A, Iwashita Y, Shibata K, et al. Improved long-term survival after liver resection for hepatocellular carcinoma in the modern era: retrospective study from HCV-endemic areas. World J Surg 2006; 30:1567–78
Ishii A, Lo SH. A role of tensin in skeletal muscle regeneration. Biochem J 2001; 356:734–45
Chen H, Duncan IC, Bozorgchami H, et al. Tensin1 and a previously undocumented family member, tensin2, positively regulate cell migration. Proc Natl Acad Sci USA 2002; 99:733–8
Chen H, Lo SH. Regulation of tensin-promoted cell migration by its focal adhesion binding and Src homology domain 2. Biochem J 2003; 370:1039–45
Yamashita M, Horikoshi S, Asanuma K, et al. Tensin is potentially involved in extracellular matrix production in mesangial cells. Histochem Cell Biol 2004; 121:245–54
Liotta LA, Kohn EC. The microenvironment of the tumor-host interface. Nature 2001; 411:375–9
Hundahl SA, Phillips JL, Menck HR. The National Cancer Data Base Report on poor survival of U.S. gastric carcinoma patients treated with gastrectomy: Fifth Edition American Joint Committee on Cancer Staging, proximal disease, and the “different disease” hypothesis. Cancer 2000; 88:921–32
Acknowledgements
We thank T. Shimooka, K. Ogata, M. Oda, N. Kasagi, and Y. Nakagawa for their excellent technical assistance. This work was supported in part by the following grants and foundations: CREST, Japan Science and Technology Agency (JST); Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research, grant numbers 17109013, 17591411, 17591413, 18390367, 18590333, 18659384, and 18790964; and The Ministry of Education, Culture, Sports, Science and Technology (MEXT) Grant-in-Aid for Scientific Research on Priority Area, grant number 18015039.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakashita, K., Mimori, K., Tanaka, F. et al. Prognostic Relevance of Tensin4 Expression in Human Gastric Cancer. Ann Surg Oncol 15, 2606–2613 (2008). https://doi.org/10.1245/s10434-008-9989-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-9989-8