Abstract
Approximately half of breast cancers that overexpress human epidermal growth factor receptor 2 (HER2) also express hormone receptors (HRs). Although HR positivity predicts efficacy of endocrine agents, preclinical and clinical data suggest that HER2 overexpression confers intrinsic resistance to hormonal treatment. In addition, HER2 overexpression is an independent adverse prognostic factor regardless of the hormonal status of the tumor, indicating that patients with HR+/HER2+ breast tumors might not derive a benefit from single-agent hormonal therapy. These data provided a strong rationale for exploring the targeting of both HR and HER2 signaling pathways in HR+/HER2+ breast tumors to optimize hormonal therapy and overcome resistance to anti-estrogen therapy. Results from a randomized clinical trial that combined hormonal treatment with targeted anti-HER2 therapy in postmenopausal women with HR+/HER2+ advanced breast cancer indicate that this novel dual-targeting strategy significantly improves outcomes compared with endocrine treatment alone. Nonetheless, other data suggest that it might achieve an inferior outcome compared with anti-HER2 therapy plus chemotherapy. Therefore, targeting both the HR and HER2 signaling pathways upfront might not be the most-effective therapeutic strategy in the management of HR+/HER2+ breast cancer. We discuss the clinical implication of resistance to endocrine therapy, and describe new insights into the management of HR+/HER2+ advanced breast cancer.
Key Points
-
Aromatase inhibitors are the most effective endocrine agents for the treatment of postmenopausal patients with breast tumors expressing hormonal receptors (HR+); however, not all HR-expressing tumors respond to endocrine therapies and those who respond eventually become resistant
-
Several mechanisms for resistance to hormonal therapy have been proposed, including downregulation of HR expression, HR mutations, altered expression of coregulators, and ligand-independent activation of estrogen receptor and coactivators by overexpression and/or amplification of HER2
-
HR+/HER2+ breast tumors are too aggressive to benefit from single-agent hormonal therapy; however, preclinical and recent clinical data indicate that such resistance might be overcome by inhibiting the HER2 pathway
-
Anti-ER/HER2 concurrent treatment provides significantly better outcomes in HR+/HER2+ advanced breast cancer than hormone therapy alone, but clinical data indicate that it might achieve an inferior outcome compared with anti-HER2 therapy plus chemotherapy
-
Combination chemotherapy with anti-HER2 therapy should be the first-line treatment option considered in patients with good performance status, visceral disease or rapidly progressing HR+/HER2+ breast tumors
-
Whatever approach is chosen for the treatment of HER2+ breast cancer, it should be given upfront with anti-HER2 therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jemal A et al. (2006) Cancer Statistics, 2006. CA Cancer J Clin 56: 106–130
Ferlay J et al. (2007) Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol 18: 581–592
Li CI et al. (2003) Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J Clin Oncol 21: 28–34
Osborne CK et al. (2005) Estrogen-receptor biology: continuing progress and therapeutic implications. J Clin Oncol 23: 1616–1622
Early Breast Cancer Trialists' Collaborative Group (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365: 1687–1717
Coombes RC et al. (2004) A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 350: 1081–1092
The Breast International Group 1-98 Collaborative Group (2005) A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med 353: 2747–2757
ATAC Trialists' Group (2005) Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years' adjuvant treatment for breast cancer. Lancet 365: 60–62
Jakesz R et al. (2005) Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years' adjuvant tamoxifen: combined results of ABCSG trial 8 and ARNO 95 trial. Lancet 366: 455–462
Eiermann W et al. (2001) Preoperative treatment of postmenopausal breast cancer patients with letrozole: a randomized double-blind multicenter study. Ann Oncol 12: 1527–1532
Smith IE et al. (2005) Neoadjuvant treatment of postmenopausal breast cancer with anastrozole, tamoxifen, or both in combination: the Immediate Preoperative Anastrozole, Tamoxifen, or Combined with Tamoxifen (IMPACT) Multicenter double-blind randomized trial. J Clin Oncol 23: 5108–5116
Cataliotti L et al. (2006) Comparison of anastrozole versus tamoxifen as preoperative therapy in postmenopausal women with hormone receptor-positive breast cancer. Cancer 106: 2095–2103
Bonneterre J et al. (2000) Anastrozole versus tamoxifen as first-line therapy for advanced breast cancer in 668 postmenopausal women: results of the tamoxifen or arimidex randomized group efficacy and tolerability study. J Clin Oncol 18: 3748–3757
Nabholtz JM et al. (2000) Anastrozole is superior to tamoxifen as first-line therapy for advanced breast cancer in postmenopausal women: results of a North American multicenter randomized trial. J Clin Oncol 18: 3758–3767
Mouridsen H et al. (2001) Superior efficacy of letrozole versus tamoxifen as first-line therapy for postmenopausal women with advanced breast cancer: results of a phase III study of the International Letrozole Breast Cancer Group. J Clin Oncol 19: 2596–2606
Mouridsen H et al. (2003) Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group. J Clin Oncol 21: 2101–2109
Paridaens R et al. (2003) Mature results of a randomized phase II multicenter study of exemestane versus tamoxifen as first-line hormone therapy for postmenopausal women with metastatic breast cancer. Ann Oncol 14: 1391–1398
Howell A et al. (2002) Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol 20: 3396–3403
Osborne CK et al. (2002) Double-blind, randomized trial comparing the efficacy and tolerability of fulvestrant versus anastrozole in postmenopausal women with advanced breast cancer progressing on prior endocrine therapy: results of a North American trial. J Clin Oncol 20: 3386–3395
Kaufman B et al. (2006) Trastuzumab plus anastrozole prolongs progression-free survival in postmenopausal women with HER2 positive, hormone-dependent metastatic breast cancer (MBC) [abstract #LBA2]. European Society for Medical Oncology (ESMO) Congress: 2006 29 September to 3 October; Istanbul, Turkey
Ring A et al. (2004) Mechanisms of tamoxifen resistance. Endocr Relat Cancer 11: 643–658
Massarweh S et al. (2007) Unraveling the mechanisms of endocrine resistance in breast cancer: new therapeutic opportunities. Clin Cancer Res 13: 1950–1954
Ali S et al. (2002) Endocrine-responsive breast cancer and strategies for combating resistance. Nat Rev Cancer 2: 101–112
Ali S et al. (1993) Modulation of transcriptional activation by ligand-dependent phosphorylation of the human oestrogen receptor A/B region. EMBO J 12: 1153–1160
Shou J et al. (2004) Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu crosstalk in ER/HER2-positive breast cancer. J Natl Cancer Inst 96: 926–935
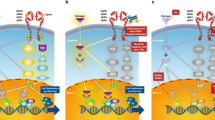
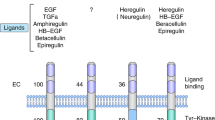
Yarden Y et al. (2001) Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2: 127–137
Hynes NE and Lane HA (2005) ErbB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5: 341–354
Benz C et al. (1992) Estrogen-dependent, tamoxifen-resistant tumorigenic growth of MCF-7 cells transfected with HER2/neu. Breast Cancer Res Treat 24: 85–95
Pietras R et al. (1995) HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene 10: 2435–2446
Schlessinger J (2004) Common and distinct elements in cellular signaling via EGF and FGF receptors. Science 306: 1506–1507
Hudziak RM et al. (1987) Increased expression of the putative growth factor receptor p185HER2 causes transformation and tumorigenesis of NIH 3T3 cells. Proc Natl Acad Sci USA 84: 7159–7163
Slamon DJ et al. (1989) Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244: 707–712
Yu D et al. (2000) Overexpression of ErbB2 in cancer and ErbB2-targeting strategies. Oncogene 11: 6115–6121
Carter P et al. (1992) Humanization of an anti-p185HER2 antibody for human cancer therapy. Proc Natl Acad Sci USA 89: 4285–4289
Tokuda Y et al. (1996) In vitro and in vivo anti-tumour effects of a humanised monoclonal antibody against c-erbB-2 product. Br J Cancer 73: 1362–1365
Baselga J et al. (1998) Recombinant humanized anti-HER2 antibody (HerceptinTM) enhances the antitumor activity of paclitaxel and doxorubicin against HER2/neu overexpressing human breast cancer xenografts. Cancer Res 58: 2825–2831
Baselga J et al. (2001) Mechanism of action of trastuzumab and scientific update. Semin Oncol 28: 4–11
Slamon DJ et al. (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344: 783–792
Marty M et al. (2005) Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: The M77001 Study Group. J Clin Oncol 23: 4265–4274
Romond EH et al. (2005) Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 353: 1673–1684
Piccart-Gebhart MJ et al. (2005) Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med 353: 1659–1672
Joensuu H et al. (2006) Adjuvant docetaxel or vinorelbine with or without trastuzumab for breast cancer. N Engl J Med 354: 809–820
Smith I (2006) Trastuzumab for early breast cancer. Lancet 367: 107
Geyer CE et al. (2006) Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 355: 2733–2743
Baselga J et al. (2007) Objective response rate in a phase II multicenter trial of pertuzumab (P), a HER2 dimerization inhibiting monoclonal antibody, in combination with trastuzumab (T) in patients (pts) with HER2-positive metastatic breast cancer (MBC) which has progressed during treatment with T [abstract #1004]. Proc Am Soc Clin Oncol: 2007 1–5 June; Chicago, IL
Beeram M et al. (2007) A phase I study of trastuzumab-MCC-DM1 (T-DM1), a first-in-class HER2 antibody-drug conjugate (ADC), in patients (pts) with HER2+ metastatic breast cancer (BC) [abstract #1042]. Proc Am Soc Clin Oncol: 2007 1–5 June; Chicago, IL
Modi S et al. (2006) Phase I trial of KOS-953, a heat shock protein 90 inhibitor, and trastuzumab (T) [abstract #501]. Proc Am Soc Clin Oncol: 2007 June 1–5; Chicago, IL
Franklin MC et al. (2004) Insights into ErbB signaling from the structure of the ErbB2-pertuzumab complex. Cancer Cell 5: 317–328
Remillard S et al. (1975) Antimitotic activity of the potent tumor inhibitor maytansine. Science 189: 1002–1005
Whitesell L et al. (2005) HSP90 and the chaperoning of cancer. Nat Rev Cancer 5: 761–772
Osborne CK et al. (2000) Selective estrogen receptor modulators: structure, function, and clinical use. J Clin Oncol 18: 3172–3186
Osborne C et al. (2001) Estrogen receptor: current understanding of its activation and modulation. Clin Cancer Res 7: 4338s–4342s
Kushner PJ et al. (2000) Estrogen receptor pathways to AP-1. J Steroid Biochem Mol Biol 74: 311–317
Nicholson RI et al. (1999) Involvement of steroid hormone and growth factor crosstalk in endocrine response in breast cancer. Endocr Relat Cancer 6: 373–387
Lee A et al. (2001) Crosstalk among estrogen receptor, epidermal growth factor, and insulin-like growth factor signaling in breast cancer. Clin Cancer Res 7: 4429s–4435s
Wilson M et al. (2002) Identification and characterization of a negative regulatory element within the epidermal growth factor receptor gene first intron in hormone-dependent breast cancer cells. J Cell Biochem 85: 601–614
Russell K et al. (1992) Transcriptional repression of the neu protooncogene by estrogen stimulated estrogen receptor. Cancer Res 52: 6624–6629
Frasor J et al. (2003) Profiling of estrogen up- and down-regulated gene expression in human breast cancer cells: insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinology 144: 4562–4574
McKenna NJ et al. (1999) Nuclear receptor coregulators: cellular and molecular biology. Endocr Rev 20: 321–344
Barnes C et al. (2004) Novel estrogen receptor coregulators and signaling molecules in human diseases. Cell Mol Life Sci 61: 281–291
Singh R et al. (2005) Steroid hormone receptor signaling in tumorigenesis. J Cell Biochem 96: 490–505
Smith CL et al. (2004) Coregulator function: a key to understanding tissue specificity of selective receptor modulators. Endocr Rev 25: 45–71
Anzick SL et al. (1997) AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science 277: 965–968
Osborne CK et al. (2003) Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. J Natl Cancer Inst 95: 353–361
Aronica S et al. (1994) Estrogen action via the cAMP signaling pathway: stimulation of adenylate cyclase and cAMP-regulated gene transcription. Proc Natl Acad Sci USA 91: 8517–8521
Kahlert S et al. (2000) Estrogen receptor alpha rapidly activates the IGF-1 receptor pathway. J Biol Chem 275: 18447–18453
Simoncini T et al. (2000) Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3-OH kinase. Nature 407: 538–541
Morelli C et al. (2003) Estrogen receptor-alpha regulates the degradation of insulin receptor substrates 1 and 2 in breast cancer cells. Oncogene 26: 4007–4016
Migliaccio A et al. (2002) Src is an initial target of sex steroid hormone action. Ann NY Acad Sci 963: 185–190
Van Cutsem E et al. (2007) Randomized phase III study of irinotecan and 5-FU/FA with or without cetuximab in the first-line treatment of patients with metastatic colorectal cancer (mCRC): The CRYSTAL trial [abstract]. J Clin Oncol 25: A4000
Song RX et al. (2002) Linkage of rapid estrogen action to MAPK activation by ERα-Shc association and Shc pathway activation. Mol Endocrinol 16: 116–127
MacGregor Schafer J et al. (2001) Estrogen receptor α mediated induction of the transforming growth factor α gene by estradiol and 4-hydroxytamoxifen in MDA-MB-231 breast cancer cells. J Steroid Biochem Mol Biol 78: 41–50
Font de Mora J et al. (2000) AIB1 is a conduit for kinase-mediated growth factor signaling to the estrogen receptor. Mol Cell Biol 20: 5041–5047
Witters L et al. (1997) Enhanced anti-proliferative activity of the combination of tamoxifen plus HER-2-neu antibody. Breast Cancer Res Treat 42: 1–5
Gutierrez MC et al. (2005) Molecular changes in tamoxifen-resistant breast cancer: relationship between estrogen receptor, HER-2, and p38 mitogen-activated protein kinase. J Clin Oncol 23: 2469–2476
Ellis MJ et al. (2001) Letrozole is more effective neoadjuvant endocrine therapy than tamoxifen for ErbB-1- and/or ErbB-2-positive, estrogen receptor-positive primary breast cancer: evidence from a phase III randomized trial. J Clin Oncol 19: 3808–3816
Johnston S et al. (2003) Aromatase inhibitors for breast cancer: lessons from the laboratory. Nat Rev Cancer 3: 821–831
Massarweh S et al. (2006) Mechanisms of tumor regression and resistance to estrogen deprivation and fulvestrant in a model of estrogen receptor-positive, HER-2/neu-positive breast cancer. Cancer Res 66: 8266–8273
Kurokawa H et al. (2000) Inhibition of HER2/neu (erbB-2) and mitogen-activated protein kinases enhances tamoxifen action against HER2-overexpressing, tamoxifen-resistant breast cancer cells. Cancer Res 60: 5887–5894
Wright C et al. (1992) Relationship between c-erbB-2 protein product expression and response to endocrine therapy in advanced breast cancer. Br J Cancer 65: 118–121
Berns E et al. (1995) Oncogene amplification and prognosis in breast cancer: relationship with systemic treatment. Gene 159: 11–18
Houston S et al. (1999) Overexpression of c-erbB2 is an independent marker of resistance to endocrine therapy in advanced breast cancer. Br J Cancer 79: 1220–1226
Lipton A et al. (2003) Serum HER-2/neu and response to the aromatase inhibitor letrozole versus tamoxifen. J Clin Oncol 21: 1967–1972
Lipton A et al. (2002) Elevated serum HER-2/neu level predicts decreased response to hormone therapy in metastatic breast cancer. J Clin Oncol 20: 1467–1472
Dowsett M et al. (2005) Biomarker changes during neoadjuvant anastrozole, tamoxifen, or the combination: influence of hormonal status and HER-2 in breast cancer—a study from the IMPACT Trialists. J Clin Oncol 23: 2477–2492
Viale G et al. (2005) Central review of ER, PgR and HER-2 in BIG 1-98 evaluating letrozole vs tamoxifen as adjuvant endocrine therapy for postmenopausal women with receptor-positive breast cancer [abstract #44]. In Proceedings of the San Antonio Breast Cancer Symposium: 9 December 2005, San Antonio, TX
Ellis MJ et al. (2006) Estrogen-independent proliferation is present in estrogen-receptor HER2-positive primary breast cancer after neoadjuvant letrozole. J Clin Oncol 24: 3019–3025
Dowsett M et al. (2006) Relationship between quantitative ER and PgR expression and HER2 status with recurrence in the ATAC trial [abstract #48]. In Proceedings of the San Antonio Breast Cancer Symposium: 2006 December 14–17; San Antonio, TX
Daly J et al. (1997) Neu differentiation factor induces ErbB2 down-regulation and apoptosis of ErbB2-overexpressing breast tumor cells. Cancer Res 57: 3804–3811
Buzdar A et al. (2001) Phase III, multicenter, double-blind, randomized study of letrozole, an aromatase inhibitor, for advanced breast cancer versus megestrol acetate. J Clin Oncol 19: 3357–3366
Buzdar A et al. (1998) Anastrozole versus megestrol acetate in the treatment of postmenopausal women with advanced breast carcinoma: results of a survival update based on a combined analysis of data from two mature phase III trials. Arimidex Study Group. Cancer 83: 1142–1152
Kaufmann M et al. (2000) Exemestane is superior to megestrol acetate after tamoxifen failure in postmenopausal women with advanced breast cancer: results of a phase III randomized double-blind trial. J Clin Oncol 18: 1399–1411
Marcom PK et al. (2007) The combination of letrozole and trastuzumab as first or second-line biological therapy produces durable responses in a subset of HER2 positive and ER positive advanced breast cancers. Breast Cancer Res Treat 102: 43–49
Kaufman B et al. (2006) Trastuzumab plus anastrozole prolongs progression-free survival in postmenopausal women with HER2-positive, hormone-dependent metastatic breast cancer [abstract #a3]. In Proceedings of the San Antonio Breast Cancer Symposium: 2006 December 14–17; San Antonio, TX
Arpino G et al. (2007) Treatment of human epidermal growth factor receptor 2-overexpressing breast cancer xenografts with multiagent HER-targeted therapy. J Natl Cancer Inst 99: 694–705
Xia W et al. (2006) A model of acquired autoresistance to a potent ErbB2 tyrosine kinase inhibitor and a therapeutic strategy to prevent its onset in breast cancer. Proc Natl Acad Sci USA 103: 7795–7800
Johnston SR (2005) Clinical trials of intracellular signal transductions inhibitors for breast cancer—a strategy to overcome endocrine resistance. Endocr Relat Cancer 12: S145–S157
Ellis M (2004) Overcoming endocrine therapy resistance by signal transduction inhibition. Oncologist 9: 20–26
Johnston SR et al. (2007) Clinical strategies for rationale combinations of aromatase inhibitors with novel therapies for breast cancer. J Steroid Biochem Mol Biol 106: 180–186
Clark AS et al. (2002) Constitutive and inducible Akt activity promotes resistance to chemotherapy, trastuzumab, or tamoxifen in breast cancer cells. Mol Cancer Ther 1: 707–717
Pancholi S et al. (2004) Elevated levels of Akt and p90rsk provide tamoxifen-resistant MCF-7 cells with a survival advantage involving Bad and Bcl-2 [abstract]. Proc Am Assoc Cancer Res 45: A5168
Baselga J et al. (2004) Phase II, 3-arm study of CCI-779 in combination with letrozole in postmenopausal women with locally advanced or metastatic breast cancer: preliminary results [abstract #544]. Proc Am Soc Clin Oncol 23
Tabernero J et al. (2005) A phase I study with tumor molecular pharmacodynamic (MPD) evaluation of dose and schedule of the oral mTOR-inhibitor everolimus (RAD001) in patients (pts) with advanced solid tumors [abstract #3007]. Proc Am Soc Clin Oncol 23
Carpenter JT et al. (2005) Randomized 3-arm, phase 2 study of temsirolimus (CCI-779) in combination with letrozole in postmenopausal women with locally advanced or metastatic breast cancer [abstract # 564]. Proc Am Soc Clin Oncol 23
O'Reilly KE et al. (2006) mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res 66: 1500–1508
Hudis CA (2007) Trastuzumab—mechanism of action and use in clinical practice. N Engl J Med 357: 39–51
Vogel CL et al. (2002) Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J Clin Oncol 20: 719–726
Burstein HJ et al. (2003) Trastuzumab and vinorelbine as first-line therapy for HER2-overexpressing metastatic breast cancer: multicenter phase II trial with clinical outcomes, analysis of serum tumor markers as predictive factors, and cardiac surveillance algorithm. J Clin Oncol 21: 2889–2895
Robert N et al. (2006) Randomized phase III study of trastuzumab, paclitaxel, and carboplatin compared with trastuzumab and paclitaxel in women with HER-2-overexpressing metastatic breast cancer. J Clin Oncol 24: 2786–2792
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Prat, A., Baselga, J. The role of hormonal therapy in the management of hormonal-receptor-positive breast cancer with co-expression of HER2. Nat Rev Clin Oncol 5, 531–542 (2008). https://doi.org/10.1038/ncponc1179
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncponc1179
This article is cited by
-
Anti-HER2 Drugs for the Treatment of Advanced HER2 Positive Breast Cancer
Current Treatment Options in Oncology (2023)
-
Effects of neoadjuvant trastuzumab, pertuzumab and palbociclib on Ki67 in HER2 and ER-positive breast cancer
npj Breast Cancer (2022)
-
Impact of hormone therapy side effects on health-related quality of life, distress, and well-being of breast cancer survivors
Scientific Reports (2022)
-
Effect of level of hormone-receptor expression on treatment outcomes of “triple-positive” early-stage breast cancer
Breast Cancer Research and Treatment (2021)
-
A preclinical PET dual-tracer imaging protocol for ER and HER2 phenotyping in breast cancer xenografts
EJNMMI Research (2020)