Abstract
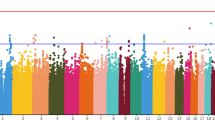
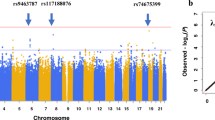
Administration of certain drugs (for example, antiarrhythmics, antihistamines, antibiotics, antipsychotics) may occasionally affect myocardial repolarization and cause prolongation of the QT interval. We performed a whole genome association study of drug-induced QT prolongation after 14 days of treatment in a phase 3 clinical trial evaluating the efficacy, safety and tolerability of a novel atypical antipsychotic, iloperidone, in patients with schizophrenia. We identified DNA polymorphisms associated with QT prolongation in six loci, including the CERKL and SLCO3A1 genes. Each single nucleotide polymorphism (SNP) defined two genotype groups associated with a low mean QT change (ranging from −0.69 to 5.67 ms depending on the SNP) or a higher mean QT prolongation (ranging from 14.16 to 17.81 ms). The CERKL protein is thought to be part of the ceramide pathway, which regulates currents conducted by various potassium channels, including the hERG channel. It is well established that inhibition of the hERG channel can prolong the QT interval. SLCO3A1 is thought to play a role in the translocation of prostaglandins, which have known cardioprotective properties, including the prevention of torsades de pointes. Our findings also point to genes involved in myocardial infarction (PALLD), cardiac structure and function (BRUNOL4) and cardiac development (NRG3). Results of this pharmacogenomic study provide new insight into the clinical response to iloperidone, developed with the goal of directing therapy to those patients with the optimal benefit/risk ratio.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Al Khatib SM, LaPointe NM, Kramer JM, Califf RM . What clinicians should know about the QT interval. JAMA 2003; 289: 2120–2127.
Paulussen ADC, Gilissen RAHJ, Armstrong M, Doevendans PA, Verhasselt P, Smeets HJM et al. Genetic variations of KCNQ1, KCNH2, SCN5A, KCNE1, and KCNE2 in drug-induced long QT syndrome patients. J Mol Med 2004; 82: 182–188.
Roden DM . Drug-induced prolongation of the QT interval. N Engl J Med 2004; 350: 1013–1022.
Zareba W, Lin DA . Antipsychotic drugs and QT interval prolongation. Psychiatr Q 2003; 74: 291–306.
Yang P, Kanki H, Drolet B, Yang T, Wei J, Viswanathan PC et al. Allelic variants in long-QT disease genes in patients with drug-associated torsades de pointes. Circulation 2002; 105: 1943–1948.
Farkas A, Coker SJ . Prevention of clofilium-induced torsades de pointes by prostaglandin E2 does not involve ATP-dependent K+ channels. Eur J Pharmacol 2003; 472: 189–196.
Cutler AJ, Kalali AH, Weiden P, Hamilton J, Wolfgang CD . Four-week, double-blind, placebo- and ziprasidone-controlled trial of iloperidone in patients with acute exacerbations of schizophrenia. J Clin Psychopharmacol 2008; 28 (Suppl 1): S20–S28.
Di X, Matsuzaki H, Webster TA, Hubbell E, Liu G, Dong S et al. Dynamic model based algorithms for screening and genotyping over 100K SNPs on oligonucleotide microarrays. Bioinformatics 2005; 21: 1958–1963.
BRLMM: an improved genotype calling method for the GeneChip® Human Mapping 500K Array Set. Affymetrix Web site. http://www.affymetrix.com/support/technical/whitepapers/brlmm_whitepaper.pdf. Accessed 26 July 2007.
Benjamini Y, Hochberg Y . Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc 1995; 57: 289–300.
Weller JI, Song JZ, Heyen DW, Lewin HA, Ron M . A new approach to the problem of multiple comparisons in the genetic dissection of complex traits. Genetics 1998; 150: 1699–1706.
Mosig MO, Lipkin E, Khutoreskaya G, Tchourzyna E, Soller M, Friedmann A . A whole genome scan for quantitative trait loci affecting milk protein percentage in Israeli-Holstein cattle, by means of selective milk DNA pooling in a daughter design, using an adjusted false discovery rate criterion. Genetics 2001; 157: 1683–1698.
Benjamini Y, Yekutieli D . Quantitative trait Loci analysis using the false discovery rate. Genetics 2005; 171: 783–790.
Barrett JC, Fry B, Maller J, Daly MJ . Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 2005; 21: 263–265.
Lewontin RC . On measures of gametic disequilibrium. Genetics 1988; 120: 849–852.
Bornancin F, Mechtcheriakova D, Stora S, Graf C, Wlachos A, Devay P et al. Characterization of a ceramide kinase-like protein. Biochim Biophys Acta 2005; 1687: 31–43.
Sherry ST, Ward MH, Kholodov M, Baker J, Phan L, Smigielski EM, Sirotkin K . dbSNP: the NCBI database of genetic variation. Nucleic Acids Res 2001; 29: 308–311.
Chapman H, Ramstrom C, Korhonen L, Laine M, Wann KT, Lindholm D et al. Downregulation of the HERG (KCNH2) K+ channel by ceramide: evidence for ubiquitin-mediated lysosomal degradation. J Cell Sci 2005; 118: 5325–5334.
Hida H, Takeda M, Soliven B . Ceramide inhibits inwardly rectifying K+ currents via a Ras- and Raf-1-dependent pathway in cultured oligodendrocytes. J Neurosci 1998; 18: 8712–8719.
Chik CL, Li B, Karpinski E, Ho AK . Ceramide inhibits the outward potassium current in rat pinealocytes. J Neurochem 2001; 79: 339–348.
Ceramide kinase-like protein. Human Protein Reference Database Web site. Available at http://www.hprd.org. Accessed 7 August 2007.
UniProtKB/Swiss-Prot entry Q49MI3. ExPASy Web Site. Available at http://www.expasy.org/cgi-bin/niceprot.pl?CERKL_HUMAN. Accessed 8 August 2007.
Adachi H, Suzuki T, Abe M, Asano N, Mizutamari H, Tanemoto M et al. Molecular characterization of human and rat organic anion transporter OATP-D. Am J Physiol Renal Physiol 2003; 285: F1188–F1197.
Thiemermann C, Zacharowski K . Selective activation of E-type prostanoid3-receptors reduces myocardial infarct size: a novel insight into the cardioprotective effects of prostaglandins. Pharmacol Ther 2000; 87: 61–67.
Ladd AN, Charlet BN, Cooper TA . The CELF family of RNA binding proteins is implicated in cell-specific and developmentally regulated alternative splicing. Mol Cell Biol 2001; 21: 1285–1296.
Lu X, Timchenko NA, Timchenko LT . Cardiac elav-type RNA-binding protein (ETR-3) binds to RNA CUG repeats expanded in myotonic dystrophy. Hum Mol Genet 1999; 8: 53–60.
Ladd AN, Taffet G, Hartley C, Kearney DL, Cooper TA . Cardiac tissue-specific repression of CELF activity disrupts alternative splicing and causes cardiomyopathy. Mol Cell Biol 2005; 25: 6267–6278.
Philips AV, Timchenko LT, Cooper TA . Disruption of splicing regulated by a CUG-binding protein in myotonic dystrophy. Science 1998; 280: 737–741.
Li D, Bachinski LL, Roberts R . Genomic organization and isoform-specific tissue expression of human NAPOR (CUGBP2) as a candidate gene for familial arrhythmogenic right ventricular dysplasia. Genomics 2001; 74: 396–401.
Ladd AN, Stenberg MG, Swanson MS, Cooper TA . Dynamic balance between activation and repression regulates pre-mRNA alternative splicing during heart development. Dev Dyn 2005; 233: 783–793.
Carteron C, Ferrer-Montiel A, Cabedo H . Characterization of a neural-specific splicing form of the human neuregulin 3 gene involved in oligodendrocyte survival. J Cell Sci 2006; 119: 898–909.
Panchal H, Wansbury O, Parry S, Ashworth A, Howard B . Neuregulin3 alters cell fate in the epidermis and mammary gland. BMC Dev Biol 2007; 7: 105.
Révillion F, Lhotellier V, Hornez L, Bonneterre J, Peyrat JP . ErbB/HER ligands in human breast cancer, and relationships with their receptors, the bio-pathological features and prognosis. Ann Oncol 2008; 19: 73–80.
Zhao Y-Y, Sawyer DR, Baliga RR, Opel DJ, Han X, Marchionni MA et al. Neuregulins promote survival and growth of cardiac myocytes: persistence of ErbB2 and ErbB4 expression in neonatal and adult ventricular myocytes. J Biol Chem 1998; 273: 10261–10269.
Shiffman D, Ellis SG, Rowland CM, Malloy MJ, Luke MM, Iakoubova OA et al. Identification of four gene variants associated with myocardial infarction. Am J Hum Genet 2005; 77: 596–605.
Jin L, Kern MJ, Otey CA, Wamhoff BR, Somlyo AV . Angiotensin II, focal adhesion kinase, and PRX1 enhance smooth muscle expression of lipoma preferred partner and its newly identified binding partner paladin to promote cell migration. Circ Res 2007; 100: 817–825.
Arking DE, Pfeufer A, Post W, Kao WHL, Newton-Cheh C, Ikeda M et al. A common genetic variant in the NOS1 regulator NOS1AP modulates cardiac repolarization. Nat Genet 2006; 38: 644–651.
Acknowledgements
We thank all the patients for participating in this study. We thank Michael Di Marino, Ingeborg Holt and Nan Wang for their assistance in the statistical analysis, and Andrew Thompson for his technical support. We also thank Dr Paolo Baroldi and Chip Clark for their critical review of the article. This study was funded and submitted by Vanda Pharmaceuticals Inc. Study design, data collection, data analysis and data interpretation were completed by the authors, who are employees of Vanda Pharmaceuticals Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volpi, S., Heaton, C., Mack, K. et al. Whole genome association study identifies polymorphisms associated with QT prolongation during iloperidone treatment of schizophrenia. Mol Psychiatry 14, 1024–1031 (2009). https://doi.org/10.1038/mp.2008.52
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2008.52
Keywords
This article is cited by
-
Delineating significant genome-wide associations of variants with antipsychotic and antidepressant treatment response: implications for clinical pharmacogenomics
Human Genomics (2020)
-
QTc prolongation in short-term treatment of schizophrenia patients: effects of different antipsychotics and genetic factors
European Archives of Psychiatry and Clinical Neuroscience (2018)
-
Clinical Association Between Pharmacogenomics and Adverse Drug Reactions
Drugs (2015)
-
Drug–gene interactions and the search for missing heritability: a cross-sectional pharmacogenomics study of the QT interval
The Pharmacogenomics Journal (2014)
-
Pharmacogenomics of adverse drug reactions
Genome Medicine (2013)