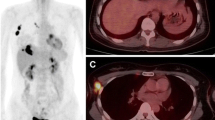
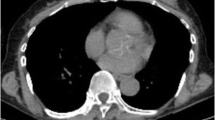
Abstract
Purpose
To evaluate the relationship between FDG uptake and prognostic factors of breast cancer such as hormone receptors (estrogen and progesterone), expression of c-erbB-2, axillary lymph node status, tumor histology, grade and size.
Materials and methods
Between May 2009 and February 2011; 79 patients (mean age ± SD: 52.9 ± 13.9 years) with biopsy proven breast cancer underwent F-18 FDG PET/CT scanning for staging. Patients with excisional biopsy or neoadjuvant chemotherapy were excluded from the study. Histological types included were invasive ductal carcinoma (n = 68), invasive lobular carcinoma (n = 2), and invasive ductal plus lobular mixed carcinoma (n = 9). Maximum standardized uptake values (SUVmax) were compared with estrogen (ER) and progesterone receptors (PR), expression of c-erbB-2, as well as tumor grade and tumor size. For the evaluation of relationship between tumor SUVmax values and prognosticators such as hormone receptors, tumor histologic grade, and tumor size, statistical analyses were performed using Student t test, Mann–Whitney U Test and Pearson correlation coefficient and p values of less than 0.05 were considered to indicate statistically significant differences.
Results
All primary breast neoplasms were detected by PET/CT scanner. The mean SUVmax values and breast cancer tumor sizes ranged from 2.09 to 39.0 and 0.7 to 10 cm, respectively. Tumors with negative ER [(n = 19); SUVmax median (min–max): 15 (2.09–39.0)] were associated with higher SUVmax values (p = 0.01). Tumors with overexpression of C-erbB-2 [(n = 28); SUVmax median (min–max): 16.0 (5.0-39.0)]; tumor grade 3 [(n = 25); SUVmax median (min–max): 15 (6.43–39)]; axillary lymph node involvement [(n = 60); SUVmax median (min–max): 13.61 (4.0–39.0)]; tumor histopathology and increased tumor size were associated with higher maximum standardized uptake values. However, PR did not show any relationship with SUVmax values.
Conclusion
In the present report, strong relationships were detected between the negativity of ER, overexpression of c-erbB-2, tumor grade, tumor size, histopathology, axillary lymph node involvement and SUVmax values. Accordingly, we believe that SUVmax values obtained with 18F-FDG PET/CT may provide some information about tumor biology of breast cancer.

Similar content being viewed by others
References
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, et al. Cancer statistics, 2005. CA Cancer J Clin. 2005;55:10–30.
Osborne JR, Port E, Gonen M, Doane A, Yeung H, Gerald W, et al. 18F-FDG PET of locally invasive breast cancer and association of estrogen receptor status with standardized uptake value: microarray and immunohistochemical analysis. J Nucl Med. 2010;51:543–50.
Dunnwald LK, Doot RK, Specht JM, Gralow JR, Ellis GK, Livingston RB, et al. PET tumor metabolism in locally advanced breast cancer patients undergoing neoadjuvant chemotherapy: value of static versus kinetic measures of fluorodeoxyglucose uptake. Clin Cancer Res. 2011;17:2400–9.
Oshida M, Uno K, Suzuki M, Nagashima T, Hashimoto H, Yagata H, et al. Predicting the prognoses of breast carcinoma patients with positron emission tomography using 2-deoxy-2-fluoro[18F]-d-glucose. Cancer. 1998;82:2227–34.
Berriolo-Riedinger A, Touzery C, Riedinger JM, Toubeau M, Coudert B, Arnould L, et al. 18F-FDG-PET predicts complete pathological response of breast cancer to neoadjuvant chemotherapy. Eur J Nucl Med Mol Imaging. 2007;34:1915–24.
Duch J, Fuster D, Muñoz M, Fernández PL, Paredes P, Fontanillas M, et al. 18F-FDG PET/CT for early prediction of response to neoadjuvant chemotherapy in breast cancer. Eur J Nucl Med Mol Imaging. 2009;36:1551–7.
Borg A, Tandon AK, Sigurdsson H, Clark GM, Fernö M, Fuqua SA, et al. HER-2/neu amplification predicts poor survival in node-positive breast cancer. Cancer Res. 1990;50:4332–7.
Knight WA, Livingston RB, Gregory EJ, McGuire WL. Estrogen receptor as an independent prognostic factor for early recurrence in breast cancer. Cancer Res. 1977;37:4669–71.
Parkes HC, Lillycrop K, Howell A, Craig RK. C-erbB2 mRNA expression in human breast tumours: comparison with c-erbB2 DNA amplification and correlation with prognosis. Br J Cancer. 1990;61:39–45.
Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, et al. American Society of Clinical Oncology 2007 update of recommendations fort the use of tumor markers in breast cancer. J Clin Oncol. 2007;25:5287–312.
Avril N, Menzel M, Dose J, Schelling M, Weber W, Jänicke F, et al. Glucose metabolism of breast cancer assessed by 18F-FDG PET: histologic and immunohistochemical tissue analysis. J Nucl Med. 2001;42:9–16.
Bos R, van Der Hoeven JJ, van Der Wall E, Bos R, van Der Hoeven JJ, van Der Wall E, et al. Biologic correlates of (18)fluorodeoxyglucose uptake in human breast cancer measured by positron emission tomography. J Clin Oncol. 2002;20:379–87.
Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991;19:403–10.
Mavi A, Cermik TF, Urhan M, Puskulcu H, Basu S, Yu JQ, et al. The effects of estrogen, progesterone, and C-erbB-2 receptor states on 18F-FDG uptake of primary breast cancer lesions. J Nucl Med. 2007;48:1266–72.
Ueda S, Tsuda H, Asakawa H, Shigekawa T, Fukatsu K, Kondo N, et al. Clinicopathological and prognostic relevance of uptake level using 18F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18F-FDG PET/CT) in primary breast cancer. Jpn J Clin Oncol. 2008;38:250–8.
Buck A, Schirrmeister H, Kühn T, Shen C, Kalker T, Kotzerke J, et al. FDG uptake in breast cancer: correlation with biological and clinical prognostic parameters. Eur J Nucl Med Mol Imaging. 2002;29:1317–23.
Heudel P, Cimarelli S, Montella A, Bouteille C, Mognetti T. Value of PET-FDG in primary breast cancer based on histopathological and immunohistochemical prognostic factors. Int J Clin Oncol. 2010;15:588–93.
Costelloe CM, Rohren EM, Madewell JE, Hamaoka T, Theriault RL, Yu TK, et al. Imaging bone metastases in breast cancer: techniques and recommendations for diagnosis. Lancet Oncol. 2009;10:606–14.
Yang WT. Staging of breast cancer with ultrasound. Semin Ultrasound CT MR. 2011;32:331–41.
Higashi T, Tamaki N, Honda T, Torizuka T, Kimura T, Inokuma T, et al. Expression of glucose transporters in human pancreatic tumors compared with increased FDG accumulation in PET study. J Nucl Med. 1997;38:1337–44.
Reske SN, Grillenberger KG, Glatting G, Port M, Hildebrandt M, Gansauge F, et al. Overexpression of glucose transporter 1 and increased FDG uptake in pancreatic carcinoma. J Nucl Med. 1997;38:1344–8.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–82.
Basu S, Chen W, Tchou J, Mavi A, Cermik T, Czerniecki B, et al. Comparison of triple-negative and estrogen receptor-positive/progesterone receptor-positive/HER2-negative breast carcinoma using quantitative fluorine-18 fluorodeoxyglucose/positron emission tomography imaging parameters: a potentially useful method for disease characterization. Cancer. 2008;112:995–1000.
Straver ME, Aukema TS, Olmos RA, Rutgers EJ, Gilhuijs KG, Schot ME, et al. Feasibility of FDG PET/CT to monitor the response of axillary lymph node metastases to neoadjuvant chemotherapy in breast cancer patients. Eur J Nucl Med. 2010;37:1069–76.
Hodgson NC, Gulenchyn KY. Is there a role for positron emission tomography in breast cancer staging? J Clin Oncol. 2008;26:712–20.
Greco M, Crippa F, Agresti R, Seregni E, Gerali A, Giovanazzi R, et al. Axillary lymph node staging in breast cancer by 2-fluoro-2-deoxy-d-glucose-positron emission tomography: clinical evaluation and alternative management. J Natl Cancer Inst. 2001;93:630–5.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sanli, Y., Kuyumcu, S., Ozkan, Z.G. et al. Increased FDG uptake in breast cancer is associated with prognostic factors. Ann Nucl Med 26, 345–350 (2012). https://doi.org/10.1007/s12149-012-0579-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-012-0579-2