Abstract
Purpose
To evaluate the impact of intermittent portal clamping (IPC) during surgery on the early recurrence of hepatocellular carcinoma (HCC).
Methods
The subjects of this retrospective study were 266 patients who underwent curative liver resection for HCC. The patients were grouped as follows: an intermittent portal clamping (IPC) group, n = 78; a continuous portal clamping (CPC) group, n = 128; and a non-portal clamping (NPC) group, n = 60.
Results
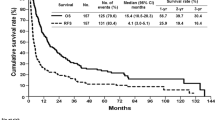
The median recurrence-free interval within 2 years of follow-up was significantly shorter in the IPC group (14.2 ± 4.6 months) than in the CPC group (18.0 ± 4.8 months, P = 0.008) or the NPC group (19.04 ± 4.1 months, P = 0.023). Moreover, 2-year recurrence-free survival was much lower in the IPC group than in the CPC group (63.6 vs. 75.8 %, P = 0.025) or the NPC group (63.6 vs. 78.0 %, P = 0.030). However, the 2-year OS rate among the three groups was comparable (72.7 vs. 79.9 %; P = 0.101) and 83.1 %, (P = 0.125). According to univariable analysis, tumor size (>5 cm), tumor number (≥2), tumor grade (low/undifferentiated), TNM stage (III), vascular infiltration, blood transfusion, and IPC were significantly associated with the early postoperative recurrence of HCC. After multivariate analysis, significance of tumor grade (low/undifferentiated) and TNM stage (III) disappeared, whereas tumor size (>5 cm), tumor number (≥2), vascular infiltration, blood transfusion, and IPC remained significant.
Conclusions
IPC is an independent risk factor for the early recurrence of HCC after surgery.


Similar content being viewed by others
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- IPC:
-
Intermittent portal clamping
- CPC:
-
Continuous portal clamping
- NPC:
-
Non-portal clamping
- RFS:
-
Recurrence-free survival
- OS:
-
Overall survival
- CI:
-
Confidence interval
- RR:
-
Risk ratio
References
Shah SA, Clearly SP, Wei AC, Yang I, Taylor BR, Hemming AW, et al. Recurrence after liver resection for hepatocellular carcinoma: risk factors, treatment, and outcomes. Surgery. 2007;141(3):330–9.
Shah SA, Greig PD, Gallinger S, Cattral MS, Dixon E, Kim RD, et al. Factors associated with early recurrence after resection for hepatocellular carcinoma and outcomes. J Am Coll Surg. 2006;202:275–83.
Cha C, Fong Y, Jarnagin WR, Blumgart LH, DeMatteo RP. Predictors and patterns of recurrence after resection of hepatocellular carcinoma. J Am Coll Surg. 2003;197(5):753–8.
Belghiti J, Noun R, Malafosse R, Mlofosse R, Jaqot P, Sauvanet A, et al. Continuous versus intermittent portal triad clamping for liver resection. Ann Surg. 1999;229:369–75.
Nuzzo G, Giuliante F, Giovannini I, Tebala GD, Cosmo G. Hepatic resections in normothermic ischemia. Surgery. 1996;120:852–8.
Imamura H, Matsuyama Y, Tanaka E, Ohkubo T, Hasegawa K, Miyagawa S, et al. Risk factors contributing to early and late phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol. 2003;38:200–7.
Katz SC, Shia J, Liau KH, Gonen M, Ruo L, Jarnagin WR, et al. Operative blood loss independently predicts recurrence and survival after resection of hepatocellular carcinoma. Ann Surg. 2009;249:617–23.
Mizuquchi T, Kawamoto M, Mequro M, Okita K, Ota S, Ishii M, Nishidate T, Furuhata T, Hirata K. Impact of aging on morbidity and mortality after liver resection: a systematic review and meta-analysis. Surg Today. 2015;45:259–70.
Poon RT, Fan ST, Wong J. Risk factors, prevention, management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232(1):10–24.
Regimbeau JM, Abdalla EK, Vauthey JN, Lauwers GY, Durand F, Nagorney DM, et al. Risk factors for early death due to recurrence after liver resection for hepatocellular carcinoma: results of a multicenter study. J Surg Oncol. 2004;85:36–41.
Tandon P, Garcia-Tsao G. Prognostic indicators in hepatocellular carcinoma: a systemic review of 72 studies. Liver Int. 2009;29:502–10.
Nanashima A, Naqayasu T. Development and clinical usefulness of the liver hanging maneuver in various anatomical hepatectomy procedures. Surg Today. 2015;. doi:10.1007/s00595-015-1166-7.
Kusano T, Sasaki A, Kai S, Endo Y, Iwaki K, Shibata K, et al. Predictors and prognostic significance of operative complications in patients with hepatocellular carcinoma who underwent hepatic resection. Eur J Surg Oncol. 2009;35:1179–85.
Hanazaki K, Kajikawa S, Shimozawa N, Mihara M, Shimada K, Hiraquri M, et al. Survival and recurrence after hepatic resection of 386 consecutive patients with hepatocellular carcinoma. J Am Coll Surg. 2000;191:381–8.
Bruns H, Kratschmer K, Hins U, Brechtel A, Keller M, Buchler MW, et al. Quality of life after curative liver resection: a single center analysis. World J Gastroenterol. 2010;16:2388–95.
Lei Z, Chang L, Fan-di M, Qi-Fei W, Ming-Hui T, Ji-Chao W. Exploration on surgical-related factors influencing HCC patients prognosis. Hepatogastroenterology. 2012;59:1541–3.
de Boer MT, Molenaar IQ, Porte RJ. Impact of blood loss on outcome after liver resection. Dig Surg. 2007;24:259–64.
Buell JF, Koffron A, Yoshida A, Hanaway M, Lo A, Layman R, et al. Is any method of vascular control superior in hepatic resection of metastatic cancers? Longmire clamping, pringle maveuver, and total vascular isolation. Arch Surg. 2001;136(5):569–75.
Doi K, Horiuchi T, Uchinami M, Tabo T, Kimura N, Yokomachi J. Hepatic ischemia–reperfusion promotes liver metastasis of colon cancer. J Surg Res. 2002;105(2):243–7.
Nicoud IB, Jones CM, Pierce JM, Earl TM, Matrisian LM, Chari RS, et al. Warm hepatic ischemia–reperfusion promotes growth of colorectal carcinoma micrometastases in mouse liver via matrix metalloproteinase-9 induction. Cancer Res. 2007;67(6):2720–8.
Man K, Ng KT, Lo CM, Ho JW, Sun BS, Sun CK, et al. Ischemia–reperfusion of small liver remnant promotes liver tumor growth and metastasis—activation of cell invasion and migration pathways. Liver Transpl. 2007;13(12):1669–77.
Nijkamp MW, van der Bilt JD, Snoeren N, Hoogwater FJ, van Houdt WJ, Molenaar IQ, et al. Prolonged portal triad clamping during liver surgery for colorectal liver metastases is associated with decreased time to hepatic tumor recurrence. Eur J Surg Oncol. 2010;36(2):182–8.
Wang J, Liu Y, Zhang A, Li C, Dong J. Inhibitory CpG sequences reduced ischemis–reperfusion-induced hepatic metastases of liver tumor in a murine model. J Surg Res. 2012;178(1):248–54.
Yang Y, Fu SY, Lau WY, Lai ECh, Li AJ, Zhou WP, et al. Selective main portal vein clamping to minimize the risk of recurrence after curative liver resection for hepatocellular carcinoma. Hepatogastroenterology. 2012;59(117):1560–5.
Ishizuka M, Kubota K, Kita J, Shimoda M, Kato M, Sawada T. Duration of hepatic vascular inflow clamping and survival after liver resection for hepatocellular carcinoma. Br J Surg. 2011;98(9):1284–90.
Yamanaka N, Okamoto E, Fujihara S, Kato T, Fujimoto J, Oriyama T, et al. Do the tumor cells of hepatocellular carcinoma dislodge into the portal venous stream during hepatic resection? Cancer. 1992;70(9):2263–7.
Matsumata T, Kanemetsu T, Takenaka K, Sugimachi K. Lack of intrahepatic recurrence of hepatocellular carcinoma by temporary portal venous embolization with starch microspheres. Surgery. 1989;105(2):188–91.
Tanaka T, Yamanaka N, Oriyama T, Furakawa K, Okamoto, E. Factors regulating tumor pressure in hepatocellular carcinoma and implication for tumor spread. Hepatology. 1997;26(2):283–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Shenghua Hao and other co-authors have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hao, S., Chen, S., Yang, X. et al. Impact of intermittent portal clamping on the early recurrence of hepatocellular carcinoma after surgery. Surg Today 46, 1290–1295 (2016). https://doi.org/10.1007/s00595-016-1316-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-016-1316-6