Abstract
Purpose
Oral mucositis induced by radiation or chemoradiation can compromise the quality of life of oral squamous cell carcinoma (OSCC) patients. The present study was designed to evaluate the preventive effects of elemental diet (ED), Elental®, on radiotherapy- or chemoradiotherapy-induced mucositis in OSCC patients.
Patients and methods
Seventy-four patients who underwent radiation (60–70 Gy) with/without chemotherapy [S-1, cisplatin (CDDP), CDDP plus S-1] were enrolled in this retrospective study; 37 had received Elental® during treatment (Elental® group) and 37 had not (control group). Factors related to alleviation of oral mucositis were identified by multivariate logistic regression analysis. Rates of completion of chemoradiation treatments were compared between Elental® and control groups according to the treatment regimen. The comparison of the nutritional status between groups was also performed.
Results
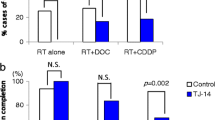
Multivariate analysis indicated that the administration of Elental® and no combined chemotherapy (radiation alone) were significant factors associated with the degree of oral mucositis, i.e., most of the patients who consumed Elental® suffered from a lower degree of mucositis compared to the control group. Elental® was associated with a significantly improved rate of completion of chemoradiation (no interruption). There was no significant difference between Elental® group and control group in terms of mean change of body weight or total protein and albumin levels in blood serum before and after (chemo)radiation.
Conclusions
The present study indicates that Elental® is effective for ameliorating oral mucositis induced by (chemo)radiation in OSCC patients. Elental® was also associated with improved completion rates of (chemo)radiotherapy.

Similar content being viewed by others
References
Rautava J, Luukkaa M, Heikinheimo K, Alin J, Grenman R, Happonen RP (2007) Squamous cell carcinomas arising from different types of oral epithelia differ in their tumor and patient characteristics and survival. Oral Oncol 43:911–919
Funk GF, Karnell LH, Robinson RA, Zhen WK, Trask DK, Hoffman HT (2002) Presentation, treatment, and outcome of oral cavity cancer: a national cancer data base report. Head Neck 24:165–180
Mehrotra R, Singh MK, Pandya S, Singh M (2008) The use of an oral brush biopsy without computer-assisted analysis in the oral lesions: a study of 94 patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 106:246–253
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Lawoyin JO, Lawoyin DO, Aderinokun G (1997) Intra-oral squamous cell carcinoma in Ibadan: a review of 90 cases. Afr J Med Med Sci 26:187–188
Sonis ST (2011) Oral mucositis. Anti-Cancer Drugs 22:607–612
Robertson C, Robertson AG, Hendry JH, Roberts SA, Slevin NJ, Duncan WB, MacDougall RH, Kerr GR, O’Sullivan B, Keane TJ (1998) Similar decreases in local tumor control are calculated for treatment protraction and for interruptions in the radiotherapy of carcinoma of the larynx in four centers. Int J Radiat Oncol Biol Phys 40:319–329
Bese NS, Hendry J, Jeremic B (2007) Effects of prolongation of overall treatment time due to unplanned interruptions during radiotherapy of different tumor sites and practical methods for compensation. Int J Radiat Oncol Biol Phys 68:654–666
Withers HR, Taylor JM, Maciejewski B (1998) The hazard of accelerated tumor clonogen repopulation during radiotherapy. Acta Oncol 27:131–146
Herrmann T, Jakubek A, Trott KR (1994) The importance of the timing of a gap in radiotherapy of squamous cell carcinomas of the head and neck. Strahlenther Onkol 170:545–549
Suwinski R, Sowa A, Rutkowski T, Wydmanski J, Tarnawski R and Maciejewski B (2003) Time factor in postoperative radiotherapy: a multivariate locoregional control analysis in 868 patients. Int J Radiat Oncol Biol Phys 56:399–412, 2003.
Russo G, Haddad R, Posner M, Machtay M (2008) Radiation treatment breaks and ulcerative mucositis in head and neck cancer. Oncologist 13:886–898
Sonis ST, Oster G, Fuchs H, Bellm L, Bradford WZ, Edelsberg J, Hayden V, Eilers J, Epstein JB, LeVeque FG, Miller C, Peterson DE, Schubert MM, Spijkervet FK, Horowitz M (2001) Oral mucositis and the clinical and economic outcomes of hematopoietic stem-cell transplantation. J Clin Oncol 19:2201–2205
Keefe DM, Schubert MM, Elting LS, Sonis ST, Epstein JB, Raber-Durlacher JE, Migliorati CA, DB MG, Hutchins RD, Peterson DE, Mucositis Study Section of the Multinational Association of Supportive Care in Cancer and the International Society for Oral Oncology (2007) Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer 109:820–831
Peterson DE, Bensadoun RJ, Roila F, ESMO Guidelines Working Group (2009) Management of oral and gastrointestinal mucositis: ESMO clinical recommendations. Ann Oncol 20(Suppl 4):174–177
Quinn B, Potting CM, Stone R, Blijlevens NM, Fliedner M, Margulies A, Sharp L (2008) Guidelines for the assessment of oral mucositis in adult chemotherapy, radiotherapy and haematopoietic stem cell transplant patients. Eur J Cancer 44:61–72
Henke M, Alfonsi M, Foa P, Giralt J, Bardet E, Cerezo L, Salzwimmer M, Lizambri R, Emmerson L, Chen MG, Berger D (2011) Palifermin decreases severe oral mucositis of patients undergoing postoperative radiochemotherapy for head and neck cancer: a randomized, placebo-controlled trial. J Clin Oncol 29:2815–2820
Bensinger W, Schubert M, Ang KK, Brizel D, Brown E, Eilers JG, Elting L, Mittal BB, Schattner MA, Spielberger R, Treister NS, Trotti 3rd AM (2008) NCCN Task Force Report. Prevention and management of mucositis in cancer care. J Natl Compr Canc Netw 6(Suppl 1):S1–21 quiz S22–24
Svanberg A, Ohrn K, Birgegard G (2010) Oral cryotherapy reduces mucositis and improves nutrition—a randomised controlled trial. J Clin Nurs 19:2146–2151
Scully C, Epstein J, Sonis S (2004) Oral mucositis: a challenging complication of radiotherapy, chemotherapy, and radiochemotherapy. Part 2: diagnosis and management of mucositis. Head Neck 26:77–84
Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M, Faucher C, Franquin JC (1997) Low energy helium-neon laser in the prevention of oral mucositis in patients undergoing bone marrow transplant: results of a double blind randomized trial. Int J Radiat Oncol Biol Phys 38:697–703
Online Ajinomoto Products Information, Elental®. http://www.ajinomoto.com/en/aboutus/history/chronicle_2014/09.html
Ikeura T, Takaoka M, Uchida K, Miyoshi H, Okazaki K (2014) Beneficial effect of low-fat elemental diet therapy on pain in chronic pancreatitis. Int J Chronic Dis:1–5, Article ID 862091.
Yamamoto T, Nakahigashi M, Umegae S, Kitagawa T, Matsumoto K (2006) Acute duodenal Crohn’s disease successfully managed with low-speed elemental diet infusion via nasogastric tube: a case report. World J Gastroenterol 12:649–651
Yamamoto T, Nakahigashi M, Umegae S, Kitagawa T, Matsumoto K (2005) Impact of elemental diet on mucosal inflammation in patients with active Crohn’s disease: cytokine production and endoscopic and histological findings. Inflamm Bowel Dis 11:580–588
Nakayama G, Morioka D, Murakami T, Takakura H, Miura Y, Togo S (2012) Chylous ascites occurring after low anterior resection of the rectum successfully treated with an oral fat-free elemental diet (Elental(®)). Clin J Gastroenterol 5:216–219
Choi K, Lee SS, Oh SJ, Lim SY, Lim SY, Jeon WK, Oh TY, Kim JW (2007) The effect of oral glutamine on 5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by intestinal permeability test. Clin Nutr 26:57–62
Savarese DM, Savy G, Vahdat L, Wischmeyer PE, Corey B (2003) Prevention of chemotherapy and radiation toxicity with glutamine. Cancer Treat Rev 29:501–513
Shou J, Lieberman MD, Hofmann K, Leon P, Redmond HP, Davies H, Daly JM (1991) Dietary manipulation of methotrexate-induced enterocolitis. JPEN J Parenter Enteral Nutr 15:307–312
Ziegler TR, Young LS, Benfell K, Scheltinga M, Hortos K, Bye R, Morrow FD, Jacobs DO, Smith RJ, Antin JH, Wilmore DW (1992) Clinical and metabolic efficacy of glutamine-supplemented parenteral nutrition after bone marrow transplantation. A randomized, double-blind, controlled study. Ann Intern Med 116:821–828
Ogata Y, Takeuchi M, Ishibashi N, Kibe S, Takahashi K, Uchida S, Murakami N, Yahara T, Shirouzu K (2012) Efficacy of Elental on prevention for chemotherapy-induced oral mucositis in colorectal cancer patients. Gan To Kagaku Ryoho 39:583–587 Japanese
Fukui T, Itoh Y, Orihara M, Yoshizawa K, Takeda H, Kawada S, Yoshioka T (2011) Elental prevented and reduced oral mucositis during chemotherapy in patients esophageal cancer. Gan To Kagaku Ryoho 38:2597–2601 Japanese
Online Nestle Products Information, VIVONEX® T.E.N. http://www.nestle-nutrition.com/products/Product.aspx?ProductId=a3bc4b10-3701-4020-9742-d947a30ed612
Bieri S, Bentzen SM, Huguenin P, Allal AS, Cozzi L, Landmann C, Monney M, Bernier J (2003) Early morbidity after radiotherapy with or without chemotherapy in advanced head and neck cancer. Experience from four non-randomized studies. Strahlenther Onkol 179:390–395
Bernier J, Domenge C, Ozsahin M, Matuszewska K, Lefèbvre JL, Greiner RH, Giralt J, Maingon P, Rolland F, Bolla M, Cognetti F, Bourhis J, Kirkpatrick A, Van Glabbeke M, European Organization for Research and Treatment of Cancer Trial 22931 (2004) Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N Engl J Med 350:1945–1952
Cooper JS, Pajak TF, Forastiere AA, Jacobs J, Campbell BH, Saxman SB, Kish JA, Kim HE, Cmelak AJ, Rotman M, Machtay M, Ensley JF, Chao KS, Schultz CJ, Lee N, Fu KK, Radiation Therapy Oncology Group 9501/Intergroup (2004) Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med 350:1937–1944
Lin A, Jabbari S, Worden FP, Bradford CR, Chepeha DB, Teknos TN, Liao JJ, Nyquist GG, Tsien C, Schipper MJ, Urba S, Wolf GT, Eisbruch A (2005) Metabolic abnormalities associated with weight loss during chemoirradiation of head-and-neck cancer. Int J Radiat Oncol Biol Phys 63:1413–1418
Gibson RJ, Keefe DM, Lalla RV, Bateman E, Blijlevens N, Fijlstra M, King EE, Stringer AM, Van der Velden WJ, Yazbeck R, Elad S, Bowen JM, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) (2013) Systematic review of agents for the management of gastrointestinal mucositis in cancer patients. Support Care Cancer 21:313–326
Hensley ML, Hagerty KL, Kewalramani T, Green DM, Meropol NJ, Wasserman TH, Cohen GI, Emami B, Gradishar WJ, Mitchell RB, Thigpen JT, Trotti 3rd A, Von Hoff D, Schuchter LM (2009) American Society of Clinical Oncology 2008 clinical practice guideline update: use of chemotherapy and radiation therapy protectants. J Clin Oncol 27:127–145
Peterson DE, Bensadoun RJ, Roila F, ESMO Guidelines Working Group (2011) Management of oral and gastrointestinal mucositis: ESMO clinical practice guidelines. Ann Oncol 22:vi78–vi84
Le QT, Kim HE, Schneider CJ, Muraközy G, Skladowski K, Reinisch S, Chen Y, Hickey M, Mo M, Chen MG, Berger D, Lizambri R, Henke M (2011) Palifermin reduces severe mucositis in definitive chemoradiotherapy of locally advanced head and neck cancer: a randomized, placebo-controlled study. J Clin Oncol 29:2808–2814
Bossi P, Locati LD, Licitra L (2012) Palifermin in prevention of head and neck cancer radiation-induced mucositis: not yet a definitive word on safety and efficacy profile. J Clin Oncol 30:565–567
Conflict of interest
There is no financial support for the present study, and the authors declare that there are no competing interests. The authors are fully responsible for the content of this paper. All authors reviewed and approved the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harada, K., Ferdous, T., Horinaga, D. et al. Efficacy of elemental diet on prevention for chemoradiotherapy-induced oral mucositis in patients with oral squamous cell carcinoma. Support Care Cancer 24, 953–959 (2016). https://doi.org/10.1007/s00520-015-2866-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-015-2866-7