Abstract
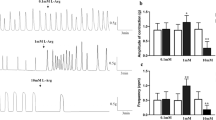
Class-I-antiarrhythmics like ajmaline are known to alter smooth muscle function, which may cause alterations in gastrointestinal motility. The effects of ajmaline on isolated gastric and portal vein smooth muscle and the underlying mechanisms are unknown. We studied the effects of ajmaline on the contractile patterns of isolated preparations of gastric antrum and portal vein from Wistar rats. The organ bath technique was used to measure spontaneous or pharmacologically induced isometric contractions. Changes in force observed after application of ajmaline or under control conditions are reported as % of the amplitude of an initial K+-induced contraction. Electric field stimulation was used to study neurogenic relaxations of gastric fundus smooth muscle. Ajmaline increased the amplitude of spontaneous contractions of muscle strips (portal vein: control 31.1 ± 15.2%, with 100 μM ajmaline 76.6 ± 32.3%, n = 9, p < 0.01; gastric antrum: control 9.5 ± 1.6%, with 100 μM ajmaline 63.9 ± 9.96%, n = 14, p < 0.01). The frequency of spontaneous activity was reduced in portal vein, but not in gastric antrum strips. The effects of ajmaline were not blocked by tetrodotoxin, L-nitroarginine methyl ester, or atropine. Ajmaline abolished coordinated neurogenic relaxations triggered by electric field stimulation and partly reversed the inhibition of GA spontaneous activity caused by the gap junction blocker carbenoxolone. Ajmaline enhances the amplitude of spontaneous contractions in rat gastric and portal vein smooth muscle. This effect may be accompanied, but not caused by an inhibition of enteric neurotransmission. Enhanced syncytial coupling as indicated by its ability to antagonize the effects of carbenoxolone is likely to underlie the enhancement of contractility.







Similar content being viewed by others
References
Bashashati M, McCallum RW (2015) Is interstitial cells of cajalopathy present in gastroparesis? J Neurogastroenterol Motil 21(4):486–493. https://doi.org/10.5056/jnm15075
Bebarova M, Matejovic P, Pasek M et al (2005) Effect of ajmaline on action potential and ionic currents in rat ventricular myocytes. Gen Physiol Biophys 24(3):311–325
Benham CD, Bolton TB, Lang RJ, Takewaki T (1986) Calcium-activated potassium channels in single smooth muscle cells of rabbit jejunum and guinea-pig mesenteric artery. J Physiol 371:45–67
Biamino G, Wessel HJ, Noring J (1975) Ajmaline-induced changes in mechanical and electrical activity of vascular smooth muscle. Blood Vessels 12(1):68–80
Bolton TB, Gordienko DV, Povstyan OV, Harhun MI, Pucovsky V (2004) Smooth muscle cells and interstitial cells of blood vessels. Cell Calcium 35(6):643–657. https://doi.org/10.1016/j.ceca.2004.01.018
Breyne J, Vanheel BJ (2004) Role of Ba2+-resistant K+ channels in endothelium-dependent hyperpolarization of rat small mesenteric arteries. Can J Physiol Pharmacol 82(1):65–71. https://doi.org/10.1139/y03-132
Chatterjee ML, De MS (1963) Pharmacological action of ajmaline, the possible mechanism of its antiarrhythmic action, and its therapeutic possibilities. Nature 200:1067–1068
D'Amato M, Curro D, Ciabattoni G et al (1990) Is peptide histidine isoleucine an inhibitory nonadrenergic noncholinergic neurotransmitter in the rat gastric fundus? Arch Int Pharmacodyn Ther 303:216–231
Echt DS, Liebson PR, Mitchell LB et al (1991) Mortality and morbidity in patients receiving encainide, flecainide, or placebo. N Engl J Med 324(12):781–788. https://doi.org/10.1056/NEJM199103213241201
Ferrari M, Furlanut M, Maragno I (1972) Effects of quinidine and ajmaline on the mechanical and electrical activity of smooth muscle. Arch Int Pharmacodyn Ther 200(1):64–69
Fischer F, Vonderlin N, Zitron E et al (2013) Inhibition of cardiac Kv1.5 and Kv4.3 potassium channels by the class Ia anti-arrhythmic ajmaline: mode of action. Naunyn Schmiedeberg’s Arch Pharmacol 386(11):991–999. https://doi.org/10.1007/s00210-013-0901-0
Friedrich O, Wegner F, Wink M et al (2007) Na+- and K+-channels as molecular targets of the alkaloid ajmaline in skeletal muscle fibres. Br J Pharmacol 151(1):82–93. https://doi.org/10.1038/sj.bjp.0707194
Gentzkow GD, Sullivan JY (1984) Extracardiac adverse effects of flecainide. Am J Cardiol 53(5):101B–105B
Golenhofen K, von Loh D (1970) Electrophysiology studies on normal spontaneous activity of the isolated guinea pig taenia coli (Elektrophysiologische Untersuchungen zur normalen Spontanaktivitat der isolierten Taenia coli des Meerschweinchens). Pflugers Arch 314(4):312–328
Hermsmeyer K (1973) Multiple pacemaker sites in spontaneously active vascular muscle. Circ Res 33(2):244–251
Hirst GDS, Garcia-Londono AP, Edwards FR (2006) Propagation of slow waves in the guinea-pig gastric antrum. J Physiol 571(Pt 1):165–177. https://doi.org/10.1113/jphysiol.2005.100735
Huizinga JD, Liu LW, Blennerhassett MG et al (1992) Intercellular communication in smooth muscle. Experientia 48(10):932–941
Khodorov BI, Zaborovskaya LD (1986) Use-dependent blockade of sodium channels by local anaesthetics and antiarrhythmic drugs. Effects of chloramine-T and calcium ions. Drugs Exp Clin Res 12(9–10):743–752
Kiesecker C, Zitron E, Lück S et al (2004) Class Ia anti-arrhythmic drug ajmaline blocks HERG potassium channels: mode of action. Naunyn Schmiedeberg’s Arch Pharmacol 370(6):423–435. https://doi.org/10.1007/s00210-004-0976-8
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG (2010) Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol 8(6):e1000412. https://doi.org/10.1371/journal.pbio.1000412
Kleinsorge H, Gaida P (1962) Behavior of the serum level after intravenous injections of ajmaline. Klin Wochenschr 40:149–151
Kohlhardt M (1984) Block of sodium currents by antiarrhythmic agents: analysis of the electrophysiologic effects of propafenone in heart muscle. Am J Cardiol 54(9):13D–19D
Koppel C, Oberdisse U, Heinemeyer G (1990) Clinical course and outcome in class IC antiarrhythmic overdose. J Toxicol Clin Toxicol 28(4):433–444
Körper S, Wink M, Fink RH (1998) Differential effects of alkaloids on sodium currents of isolated single skeletal muscle fibers. FEBS Lett 436(2):251–255
Ladabaum U, Hasler WL (1999) Motility of the small intestine. Curr Opin Gastroenterol 15(2):125–131
Lammel E, Deitmer P, Noack T (1991) Suppression of steady membrane currents by acetylcholine in single smooth muscle cells of the guinea-pig gastric fundus. J Physiol 432:259–282
Lang IM (2016) The role of central and enteric nervous Systems in the control of the retrograde giant contraction. J Neurogastroenterol Motil 22(2):321–332. https://doi.org/10.5056/jnm15141
Li X-T, Li X-Q, Hu X-M, Qiu XY (2015) The inhibitory effects of Ca2+ channel blocker nifedipine on rat Kv2.1 potassium channels. PLoS One 10(4):e0124602. https://doi.org/10.1371/journal.pone.0124602
Meinertz T, Zehender MK, Geibel A et al (1984) Long-term antiarrhythmic therapy with flecainide. Am J Cardiol 54(1):91–96
Meissner A, Noack T (2008) Proliferation of human lens epithelial cells (HLE-B3) is inhibited by blocking of voltage-gated calcium channels. Pflugers Arch 457(1):47–59. https://doi.org/10.1007/s00424-008-0514-5
Naccarelli GV, Wolbrette DL, Khan M et al (2003) Old and new antiarrhythmic drugs for converting and maintaining sinus rhythm in atrial fibrillation: comparative efficacy and results of trials. Am J Cardiol 91(6A):15D–26D
Nguyen LA, Snape WJ Jr (2015) Clinical presentation and pathophysiology of gastroparesis. Gastroenterol Clin N Am 44(1):21–30. https://doi.org/10.1016/j.gtc.2014.11.003
Noack T, Deitmer P, Golenhofen K (1990) Features of a calcium independent, caffeine sensitive outward current in single smooth muscle cells from guinea pig protal vein. Pflugers Arch 416(4):467–469
Noack T, Deitmer P, Lammel E (1992) Characterization of membrane currents in single smooth muscle cells from the guinea-pig gastric antrum. J Physiol 451:387–417
Noack T, Edwards G, Deitmer P, Greengrass P, Morita T, Andersson PO, Criddle D, Wyllie MG, Weston AH (1992) The involvement of potassium channels in the action of ciclazindol in rat portal vein. Br J Pharmacol 106(1):17–24
Oh JH, Pasricha PJ (2013) Recent advances in the pathophysiology and treatment of gastroparesis. J Neurogastroenterol Motil 19(1):18–24. https://doi.org/10.5056/jnm.2013.19.1.18
Palani D, Ghildyal P, Manchanda R (2006) Effects of carbenoxolone on syncytial electrical properties and junction potentials of guinea-pig vas deferens. Naunyn Schmiedeberg’s Arch Pharmacol 374(3):207–214. https://doi.org/10.1007/s00210-006-0109-7
Patejdl R, Noack T (2010) The propagation of excitation in portal vein smooth muscle—evidence for coupled “hot spots”. Trace Elem Electrolytes 27(4):202–208
Patejdl R, Leroux A-C, Noack T (2015) Phenytoin inhibits contractions of rat gastrointestinal and portal vein smooth muscle by inhibiting calcium entry. Neurogastroenterol Motil 27(10):1453–1465. https://doi.org/10.1111/nmo.12645
Rey E, Choung RS, Schleck CD et al (2012) Prevalence of hidden gastroparesis in the community: the gastroparesis “iceberg”. J Neurogastroenterol Motil 18(1):34–42. https://doi.org/10.5056/jnm.2012.18.1.34
Rimbäck G, Cassuto J, Faxén A et al (1986) Effect of intra-abdominal bupivacaine instillation on postoperative colonic motility. Gut 27(2):170–175
Sage D, Salin P, Alcaraz G, Castets F, Giraud P, Crest M, Mazet B, Clerc N (2007) Na(v)1.7 and Na(v)1.3 are the only tetrodotoxin-sensitive sodium channels expressed by the adult guinea pig enteric nervous system. J Comp Neurol 504(4):363–378. https://doi.org/10.1002/cne.21450
Scholz A (2002) Mechanisms of (local) anaesthetics on voltage-gated sodium and other ion channels. Br J Anaesth 89(1):52–61. https://doi.org/10.1093/bja/aef163
Sircuta C, Lazar A, Azamfirei L et al (2016) Correlation between the increased release of catecholamines evoked by local anesthetics and their analgesic and adverse effects: role of K(+) channel inhibition. Brain Res Bull 124:21–26. https://doi.org/10.1016/j.brainresbull.2016.03.009
Stevens RJ, Weinert JS, Publicover NG (1999) Visualization of origins and propagation of excitation in canine gastric smooth muscle. Am J Phys 277(3 Pt 1):C448–C460
Syrbu IF, Sokolov IS, Ozerov VN (1979) Profilaktika posleoperatsionnogo pareza zheludka i kishechnika (Prevention of postoperative gastric and intestinal paresis). Khirurgiia (Mosk) 10:107–108
Takahashi T, Owyang C (1997) Characterization of vagal pathways mediating gastric accommodation reflex in rats. J Physiol 504(Pt 2):479–488
Teo KK, Yusuf S, Furberg CD (1993) Effects of prophylactic antiarrhythmic drug therapy in acute myocardial infarction. An overview of results from randomized controlled trials. JAMA 270(13):1589–1595
van Helden DF, Laver DR, Holdsworth J, Imtiaz MS (2010) Generation and propagation of gastric slow waves. Clin Exp Pharmacol Physiol 37(4):516–524. https://doi.org/10.1111/j.1440-1681.2009.05331.x
Wolff M, Schnöbel-Ehehalt R, Mühling J et al (2014) Mechanisms of lidocaine’s action on subtypes of spinal dorsal horn neurons subject to the diverse roles of Na(+) and K(+) channels in action potential generation. Anesth Analg 119(2):463–470. https://doi.org/10.1213/ANE.0000000000000280
Zaborovskaia LD, Khodorov BI (1982) Razlichie v blokruishchem deĭstvii benzokaina i aminnykh soedineniĭ na modifitsirovannye batrakhotoksinom natrievye kanaly perekhvata Ranv’e (Differences in the blocking action of benzocaine and amino compounds on batrachotoxin-modified node of Ranvier sodium channels). Neirofiziologiia 14(6):636–643
Zhang S, Zhou Z, Gong Q et al (1999) Mechanism of block and identification of the verapamil binding domain to HERG potassium channels. Circ Res 84(9):989–998
Funding
The resources used for this work were provided by the University of Rostock. No additional funding was used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Robert Patejdl, Alina Gromann, Dietmar Bänsch, and Thomas Noack declare that they have no conflicts of interest or competing interests related to this work.
Dietmar Bänsch received research grants from Biosense, Biotronik, Medtronic, National Heart Foundation Mayo Clinic, and Zoll Medical.
Dietmar Bänsch received honoraria for lectures and consultancy from Biotronik, Bayer Medical, and Zoll Medical.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1072 kb)
Rights and permissions
About this article
Cite this article
Patejdl, R., Gromann, A., Bänsch, D. et al. Effects of ajmaline on contraction patterns of isolated rat gastric antrum and portal vein smooth muscle strips and on neurogenic relaxations of gastric fundus. Pflugers Arch - Eur J Physiol 471, 995–1005 (2019). https://doi.org/10.1007/s00424-019-02279-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-019-02279-y