Abstract
Introduction
Perforated colorectal malignancy is associated with numerous peri-operative complications and dismal long-term survival. The study aimed to review the outcome and factors predicting peri-operative complications and long-term survival of patients who underwent surgery for perforated colorectal malignancy.
Methods
A retrospective review of all patients who underwent operative intervention for perforated colorectal malignancy from February 2003 to April 2008 was performed. The severity of peritonitis was graded using the Mannheim peritonitis index (MPI).
Results
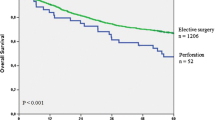
Forty-five patients, median age 67 years (36–97 years), formed the study group. Sigmoid colon (37.8%) and cecum (28.9%) were the most common sites of perforation. Sixteen (35.6%) patients had stage IV disease, while 14 (31.1%) had severe peritoneal contamination (MPI > 26). Hartmann’s procedure and right hemicolectomy were performed most frequently in 17 (37.8%) and 15 (33.4%) patients, respectively. The mortality rate in our series was 17.8%, with another 26.7% requiring surgical intensive care unit care. The independent variables predicting worse peri-operative complications were American Society of Anesthesiologists (ASA) score ≥3 and MPI >26. Left-sided perforation was the only independent factor predicting stoma creation. The only factor predicting long-term survival was the stage of malignancy (p < 0.001). The overall mean survival time for stage II, III, and IV disease were 63.7, 38.1, and 13.8 months, respectively.
Conclusions
Surgery for perforated colorectal malignancy is associated with high morbidity and mortality rates. Short-term outcome is determined by ASA score and severity of peritonitis, while long-term outcome is determined by staging of the cancer.

Similar content being viewed by others
References
Carraro PG, Segala M, Orlotti C, Tiberio G (1998) Outcome of large-bowel perforation in patients with colorectal cancer. Dis Colon Rectum 41(11):1421–1426
Lee IK, Sung NY, Lee YS, Lee SC, Kang WK, Cho HM, Ahn CH, Lee do S, Oh ST, Kim JG, Jeon HM, Chang SK (2007) The survival rate and prognostic factors in 26 perforated colorectal cancer patients. Int J Colorectal Dis 22(5):467–473
Anwar MA, D’Souza F, Coulter R, Memon B, Khan IM, Memon MA (2006) Outcome of acutely perforated colorectal cancers: experience of a single district general hospital. Surg Oncol 15(2):91–96
Alvarez JA, Baldonedo RF, Bear IG, Truán N, Pire G, Alvarez P (2005) Presentation, treatment, and multivariate analysis of risk factors for obstructive and perforative colorectal carcinoma. Am J Surg 190(3):376–382
Chen HS, Sheen-Chen SM (2000) Obstruction and perforation in colorectal adenocarcinoma: an analysis of prognosis and current trends. Surgery 127(4):370–376
Mandava N, Kumar S, Pizzi WF, Aprile IJ (1996) Perforated colorectal carcinomas. Am J Surg 172(3):236–238
Billing A, Fröhlich D, Schildberg FW (1994) Prediction of outcome using the Mannheim peritonitis index in 2003 patients. Peritonitis Study Group. Br J Surg 81(2):209–213
Astler VB, Coller FA (1954) The prognostic significance of direct extension of carcinoma of the colon and rectum. Ann Surg 139(6):846–852
Clavien PA, Sanabria JR, Mentha G, Borst F, Buhler L, Roche B, Cywes R, Tibshirani R, Rohner A, Strasberg SM (1992) Recent results of elective open cholecystectomy in a North American and a European center. Comparison of complications and risk factors. Ann Surg 216(6):618–626
Clavien PA, Camargo CA Jr, Croxford R, Langer B, Levy GA, Greig PD (1994) Definition and classification of negative outcomes in solid organ transplantation. Application in liver transplantation. Ann Surg 220(2):109–120
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Kriwanek S, Armbruster C, Dittrich K, Beckerhinn P (1996) Perforated colorectal cancer. Dis Colon Rectum 39:1409–1414
Shinkawa H, Yasuhara H, Naka S, Yanagie H, Nojiri T, Furuya Y, Ariki K, Niwa H (2003) Factors affecting the early mortality of patients with nontraumatic colorectal perforation. Surg Today 33(1):13–17
Tan KK, Zhang J, Liu JZ, Shen SF, Earnest A, Sim R (2009) Right colonic perforation in an Asian population: predictors of morbidity and mortality. J Gastrointest Surg 13(12):2252–2259
Khan S, Pawlak SE, Eggenberger JC, Lee CS, Szilagy EJ, Margolin DA (2001) Acute colonic perforation associated with colorectal cancer. Am Surg 67(3):261–264
Welch JP, Donaldson GA (1974) Perforative carcinoma of colon and rectum. Ann Surg 180(5):734–740
Veyrie N, Ata T, Muscari F, Couchard AC, Msika S, Hay JM, Fingerhut A, Dziri C (2007) Anastomotic leakage after elective right versus left colectomy for cancer: prevalence and independent risk factors. J Am Coll Surg 205(6):785–793
Konishi T, Watanabe T, Kishimoto J, Nagawa H (2006) Risk factors for anastomotic leakage after surgery for colorectal cancer: results of prospective surveillance. J Am Coll Surg 202(3):439–444
Lipska MA, Bissett IP, Parry BR, Merrie AE (2006) Anastomotic leakage after lower gastrointestinal anastomosis: men are at a higher risk. ANZ J Surg 76(7):579–585
Duchesne JC, Wang YZ, Weintraub SL, Boyle M, Hunt JP (2002) Stoma complications: a multivariate analysis. Am Surg 68:961–966
Leong QM, Koh DC, Ho CK (2008) Emergency Hartmann’s procedure: morbidity, mortality and reversal rates among Asians. Tech Coloproctol 12(1):21–25
Arumugam PJ, Bevan L, Macdonald L, Watkins AJ, Morgan AR, Beynon J, Carr ND (2003) A prospective audit of stomas—analysis of risk factors and complications and their management. Colorectal Dis 5(1):49–52
David GG, Al-Sarira AA, Willmott S, Cade D, Corless DJ, Slavin JP (2009) Use of Hartmann’s procedure in England. Colorectal Dis 11(3):308–312
Tan KK, Sim R (2010) Surgery for obstructed colorectal malignancy in an Asian population: predictors of morbidity and comparison between left and right sided cancers. J Gastrointest Surg 14(2):295–302
Steinberg SM, Barkin JS, Kaplan RS, Stablein DM (1986) Prognostic indicators of colon tumors. The Gastrointestinal Tumor Study Group experience. Cancer 57(9):1866–1870
Runkel NS, Schlag P, Schwarz V et al (1991) Outcome after emergency surgery for cancer of the large intestine. Br J Surg 78:183–188
Griffin MR, Bergstralh EJ, Coffey RJ et al (1987) Predictors of survival after curative resection of carcinoma of the colon and rectum. Cancer 60:2318–2324
Kagda FH, Nyam DC, Ho YH, Eu KW, Leong AF, Seow-Choen F (1999) Surgery may be curative for patients with a localized perforation of rectal carcinoma. Br J Surg 86(11):1448–1450
Figueredo A, Coombes ME, Mukherjee S (2008) Adjuvant therapy for completely resected stage II colon cancer. Cochrane Database Syst Rev (3):CD005390
Figueredo A, Germond C, Maroun J, Browman G, Walker-Dilks C, Wong S (1997) Adjuvant therapy for stage II colon cancer after complete resection. Provincial Gastrointestinal Disease Site Group. Cancer Prev Control 1(5):379–392
Fahy BN, D’Angelica M, DeMatteo RP, Blumgart LH, Weiser MR, Ostrovnaya I, Gonen M, Jarnagin WR (2009) Synchronous hepatic metastases from colon cancer: changing treatment strategies and results of surgical intervention. Ann Surg Oncol 16(2):361–370
Shah SA, Haddad R, Al-Sukhni W, Kim RD, Greig PD, Grant DR, Taylor BR, Langer B, Gallinger S, Wei AC (2006) Surgical resection of hepatic and pulmonary metastases from colorectal carcinoma. J Am Coll Surg 202(3):468–475
Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA (2004) Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 239(6):818–825
Scheele J, Stangl R, Altendorf-Hofmann A (1990) Hepatic metastases from colorectal carcinoma: impact of surgical resection on the natural history. Br J Surg 77(11):1241–1246
Kemeny MM, Adak S, Gray B, Macdonald JS, Smith T, Lipsitz S, Sigurdson ER, O’Dwyer PJ, Benson AB 3rd (2002) Combined-modality treatment for resectable metastatic colorectal carcinoma to the liver: surgical resection of hepatic metastases in combination with continuous infusion of chemotherapy—an intergroup study. J Clin Oncol 20(6):1499–1505
Adam R, Delvart V, Pascal G, Valeanu A, Castaing D, Azoulay D, Giacchetti S, Paule B, Kunstlinger F, Ghémard O, Levi F, Bismuth H (2004) Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg 240(4):644–657
Sugarbaker PH (2006) New standard of care for appendiceal epithelial neoplasms and pseudomyxoma peritonei syndrome? Lancet Oncol 7(1):69–76
Yan TD, Black D, Savady R, Sugarbaker PH (2006) Systematic review on the efficacy of cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for peritoneal carcinomatosis from colorectal carcinoma. J Clin Oncol 24(24):4011–4019
Verwaal VJ, Bruin S, Boot H, van Slooten G, van Tinteren H (2008) 8-year follow-up of randomized trial: cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann Surg Oncol 15(9):2426–2432
Verwaal VJ, Boot H, Aleman BM, van Tinteren H, Zoetmulder FA (2004) Recurrences after peritoneal carcinomatosis of colorectal origin treated by cytoreduction and hyperthermic intraperitoneal chemotherapy: location, treatment, and outcome. Ann Surg Oncol 11(4):375–379
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, KK., Hong, CC., Zhang, J. et al. Surgery for perforated colorectal malignancy in an Asian population: an institution’s experience over 5 years. Int J Colorectal Dis 25, 989–995 (2010). https://doi.org/10.1007/s00384-010-0945-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-010-0945-2