Abstract
Purpose
Intestinal mucositis (IM) is a common side effect of anticancer agents. Despite polychemotherapy use in clinical practice, the pathogenesis of IM has been investigated in single drug injection animal models. However, the progression of IM could vary according to drug regimens. Thus, we aimed to develop a new experimental mucositis model induced by combining irinotecan and 5-fluorouracil (5-FU) treatments.
Methods
IM was induced in male C57BL/6 mice by the intraperitoneal administration of either 0.9 % saline (5 mL/kg), irinotecan (IRI, 30 or 45 mg/kg), 5-FU (25, 37.5, or 50 mg/kg), or the combination of these doses (IRI + 5-FU) for 4 days. Animal survival, body mass variation, and diarrhea scores were evaluated daily. On the 7th day, the mice were euthanized, and intestinal samples were collected for histopathology and morphometric analysis, as well as for the determination of myeloperoxidase activity and cytokine dosage (TNF-α and IL-6).
Results
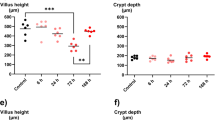
The optimal dose combination that induced IM and presented no substantial mortality on the 7th day was IRI (45 mg/kg) + 5-FU (37.5 mg/kg), which was used for subsequent studies. IRI and 5-FU in combination induced significant diarrhea, body weight loss, intestinal damage, inflammatory cell infiltration, and increased levels of cytokines when compared with other groups (P < 0.05). Neither IRI nor 5-FU alone induced IM.
Conclusions
We developed a new experimental model of IM induced by combining irinotecan and 5-FU treatments, which will allow us to gain a better knowledge concerning the pathogenesis of this disease through the pharmacological modulation of key inflammatory mediators.





Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
World Health Organization. Projections of mortality and causes of death, 2015 and 2030. http://www.who.int/healthinfo/global_burden_disease/projections/en/, accessed on June 01st, 2015
Sutcliffe SB (2012) Cancer control: life and death in an unequal world. Curr Oncol 19(1):12–15
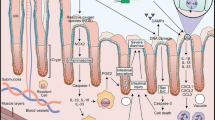
Sonis ST (2004) The pathobiology of mucositis. Nat Rev Cancer 4:277–284
Rubenstein EB, Peterson DE, Schubert M, Keefe D, Mcguire D, Epstein J, Elting LS, Fox PC, Cooksley C, Sonis ST (2004) Mucositis study section of the multinational association for supportive care in cancer; international society for oral oncology. Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer 100:2026–2046
Gibson RJ, Keefe DM, Lalla RV, Bateman E, Blijlevens N, Fijlstra M, King EE, Stringer AM, Van Der Velden WJ, Yazbeck R, Elad S, Bowen JM (2013) Mucositis study group of the multinational association of supportive care in cancer/international society of oral oncology (MASCC/ISOO). Systematic review of agents for the management of gastrointestinal mucositis in cancer patients. Support Care Cancer 21:313–326
Lee SH, Son MH, Sung KW, Choi YB, Lee NH, Yoo KH, Koo HH, do Lim H, Shin HJ (2014) Toxicity of tandem high-dose chemotherapy and autologous stem cell transplantation using carboplatin–thiotepa–etoposide and cyclophosphamide–melphalan regimens for malignant brain tumors in children and young adults. J Neurooncol 120(3):507–513
Gibson RJ, Keefe DM (2006) Cancer chemotherapy-induced diarrhea and constipation: mechanisms of damage and prevention strategies. Support Care Cancer 14:890–900
Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB (2003) The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer 98:1531–1539
Elting LS, Cooksley CD, Chambers MS, Garden AS (2007) Risk, outcomes, and costs of radiation-induced oral mucositis among patients with head-and-neck malignancies. Int J Radiat Oncol Biol Phys 68:1110–1120
Scully C, Sonis S, Diz PD (2006) Oral mucositis. Oral Dis 12:229–241
Saltz LB, Douillard JY, Pirotta N, Alakl M, Gruia G, Awad L, Elfring GL, Locker PK, Miller LL (2001) Irinotecan plus fluorouracil/leucovorin for metastatic colorectal cancer: a new survival standard. Oncologist 6:81–91
Bowen JM, Gibson RJ, Keefe DMK (2011) Animal models of mucositis: implications for therapy. J Support Oncol 9:161–168
Lalla RV (2014) Alleviating mucositis: are we on track for a novel therapeutic? Expert Rev Gastroenterol Hepatol 9(2):127–128
Melo ML, Brito GA, Soares RC, Carvalho SB, Silva JV, Soares PM, Vale ML, Souza MH, Cunha FQ, Ribeiro RA (2008) Role of cytokines (TNF-alpha, IL-1beta and KC) in the pathogenesis of CPT-11-induced intestinal mucositis in mice: effect of pentoxifylline and thalidomide. Cancer Chemother Pharmacol 61:775–784
Logan RM, Stringer AM, Bowen JM, Gibson RJ, Sonis ST, Keefe DM (2008) Serum levels of NF KappaB and drug pro-inflammatory cytokines following administration of mucotoxic drugs. Cancer BiolTher 7:1139–1145
Lima-Junior RC, Figueiredo AA, Freitas HC, Melo ML, Wong DV, Leite CA, Medeiros RP, Marques-Neto RD, Vale ML, Brito GA, Oriá RB, Souza MH, Cunha FQ, Ribeiro RA (2012) Involvement of nitric oxide on the pathogenesis of irinotecan-induced intestinal mucositis: role of cytokines on inducible nitric oxide synthase activation. Cancer Chemother Pharmacol 69:931–942
Lima-Júnior RC, Freitas HC, Wong DV, Wanderley CW, Nunes LG, Leite LL, Miranda SP, Souza MH, Brito GA, Magalhães PJ, Teixeira MM, Cunha FQ, Ribeiro RA (2014) Targeted inhibition of IL-18 attenuates irinotecan-induced intestinal mucositis in mice. Br J Pharmacol 171:2335–2350
Guabiraba R, Besnard AG, Menezes GB, Secher T, Jabir MS, Amaral SS, Braun H, Lima-Junior RC, Ribeiro RA, Cunha FQ, Teixeira MM, Beyaert R, Graham GJ, Liew FY (2014) IL-33 targeting attenuates intestinal mucositis and enhances effective tumor chemotherapy in mice. Mucosal Immunol 7:1079–1093. doi:10.1038/mi.2013.124
Soares PM, Mota JM, Gomes AS, Oliveira RB, Assreuy AM, Brito GA, Santos AA, Ribeiro RA, Souza MH (2008) Gastrointestinal dysmotility in 5-fluorouracil-induced intestinal mucositis outlasts inflammatory process resolution. Cancer Chemother Pharmacol 63:91–98
Soares PM, Lima-Junior RC, Mota JM, Justino PF, Brito GA, Ribeiro RA, Cunha FQ, Souza MH (2011) Role of platelet-activating factor in the pathogenesis of 5-fluorouracil-induced intestinal mucositis in mice. Cancer Chemother Pharmacol 68:713–720
National Institutes of Health (2011) Guide for the Care and Use of Laboratory Animals, National Academies Press (US), Washington (DC)
Kurita A, Kado S, Kaneda N, Onoue M, Hashimoto S, Yokokura T (2000) Modified irinotecan hydrochloride (CPT-11) administration schedule improves induction of delayed-onset diarrhea in rats. Cancer Chemother Pharmacol 46:211–220
Woo PCY, Ng WF, Leung HCH, Tsoi HW, Yuen KY (2000) Clarithomycin attenuates cyclophosphamide-induced mucositis in mice. Pharmacol Res 41:526–532
Alves-Filho JC, de Freitas A, Russo M, Cunha FQ (2006) Toll-like receptor 4 signaling leads to neutrophil migration impairment in polymicrobial sepsis. Crit Care Med 34:461–470
Safieh-Garabedian B, Poole S, Allchorne A, Winter J, Woolf CJ (1995) Contribution of interleukin-1 beta to the inflammation-induced increase in nerve growth factor levels and inflammatory hyperalgesia. Br J Pharmacol 115:1265–1275
Cunha FQ, Boukili MA, Motta JIB, Vargaftig BB, Ferreira SH (1993) Blockade by fenspiride of endotoxin-induced neutrophil migration in the rat. Eur J Pharmacol 238:47–52
Ballerini G, Bosi L, Castoldi GL, Ricci N (1961) Experimental enteropathy induced by 5-fluorouracil in the rat. Boll Soc Ital Biol Sper 30(37):578–580
Ikuno N, Soda H, Watanabe M, Oka M (1995) Irinotecan (CPT-11) and characteristic mucosal changes in the mouse ileum and caecum. J Natl Cancer Inst 87:1876–1888
Mashtoub S, Tran CD, Howarth GS (2013) Emu oil expedites small intestinal repair following 5-fluorouracil-induced mucositis in rats. Exp Biol Med (Maywood) 238:1305–1317
Saltz LB, Cox JV, Blanke C, Rosen LS, Fehrenbacher L, Moore MJ, Maroun JA, Ackland SP, Locker PK, Pirotta N, Elfring GL, Miller LL (2000) Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group. N Engl J Med 343:905–914
Keefe DM et al (2007) Mucositis study section of the multinational association of supportive care in cancer and the international society for oral oncology. Cancer 109:820–831
Hyatt JL, Tsurkan L, Morton CL, Yoon KJ, Harel M, Brumshtein B, Silman I, Sussman JL, Wadkins RM, Potter PM (2005) Inhibition of acetylcholinesterase by the anticancer prodrug CPT-11. Chem Biol Interact 157–158:247–252
Arifa RD, Madeira MF, de Paula TP, Lima RL, Tavares LD, Menezes-Garcia Z, Fagundes CT, Rachid MA, Ryffel B, Zamboni DS, Teixeira MM, Souza DG (2014) Inflammasome activation is reactive oxygen species dependent and mediates irinotecan-induced mucositis through IL-1β and IL-18 in mice. Am J Pathol 184:2023–2034
Wardill HR, Bowen JM, Al-Dasooqi N, Sultani M, Bateman E, Stansborough R, Shirren J, Gibson RJ (2014) Irinotecan disrupts tight junction proteins within the gut: implications for chemotherapy-induced gut toxicity. Cancer Biol Ther 15:236–244
Sakai H, Sagara A, Matsumoto K, Jo A, Hirosaki A, Takase K, Sugiyama R, Sato K, Ikegami D, Horie S, Matoba M, Narita M (2014) Neutrophil recruitment is critical for 5-fluorouracil-induced diarrhea and the decrease in aquaporins in the colon. Pharmacol Res 87:71–79
Song MK, Park MY, Sung MK (2013) 5-Fluorouracil-induced changes of intestinal integrity biomarkers in BALB/c mice. J Cancer Prev 18:322–329
Wang X, Gao J, Qian L, Gao J, Zhu S, Wu M, Zhang Y, Guan W, Ye H, Yu Y, Han W (2015) Exogenous IL-1Ra attenuates intestinal mucositis induced by oxaliplatin and 5-fluorouracil through suppression of p53-dependent apoptosis. Anticancer Drugs 26:35–45
Wong DV, Lima-Júnior RC, Carvalho CB, Borges VF, Wanderley CW, Bem AX, Leite CA, Teixeira MA, Batista GL, Silva RL, Cunha TM, Brito GA, Almeida PR, Cunha FQ, Ribeiro RA (2015) The adaptor protein Myd88 is a key signaling molecule in the pathogenesis of irinotecan-induced intestinal mucositis. PLoS ONE 10:e0139985
Frank M, Hennenberg EM, Eyking A, Rünzi M, Gerken G, Scott P, Parkhill J, Walker AW, Cario E (2015) TLR signaling modulates side effects of anticancer therapy in the small intestine. J Immunol 194(4):1983–1995
Logan RM, Stringer AM, Bowen JM, Gibson RJ, Sonis ST, Keefe DM (2009) Is the pathobiology of chemotherapy-induced alimentary tract mucositis influenced by the type of mucotoxic drug administered? Cancer Chemother Pharmacol 63:239–251
Keefe DM, Brealey J, Goland GJ, Cummins AG (2000) Chemotherapy for cancer causes apoptosis that precedes hypoplasia in crypts of the small intestine in humans. Gut 47:632–637
Cheah KY, Howarth GS, Bastian SE (2014) Grape seed extract dose-responsively decreases disease severity in a rat model of mucositis; concomitantly enhancing chemotherapeutic effectiveness in colon cancer cells. PLoS ONE 9:e85184
Lindsay RJ, Geier MS, Yazbeck R, Butler RN, Howarth GS (2010) Orally administered emu oil decreases acute inflammation and alters selected small intestinal parameters in a rat model of mucositis. Br J Nutr 104:513–519
Acknowledgments
We are grateful to Maria Silvandira Freire for her technical assistance. This study was supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Grant Nos 308879/2009-0, 307143/2014-7) and FUNCAP (Fundação Cearense de Apoio ao Desenvolvimento Científico, Grant No. 11.01.00/08). G.A.C.B., R.A.R., and R.C.P.L.J. are CNPq fellowship holders.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pereira, V.B.M., Melo, A.T., Assis-Júnior, E.M. et al. A new animal model of intestinal mucositis induced by the combination of irinotecan and 5-fluorouracil in mice. Cancer Chemother Pharmacol 77, 323–332 (2016). https://doi.org/10.1007/s00280-015-2938-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-015-2938-x